SUBMENU
Onchocerciasis is a parasitic disease caused by the filarial nematode worm Onchocerca volvulus. It is transmitted to humans by black flies (genus Simulium) and can cause severe skin and eye disease, including blindness.
The disease is commonly known as "river blindness" because the larvae of the blackfly vectors breed in fast flowing rivers. Onchocerciasis is endemic in Africa, and in 13 foci in six countries of the Americas (Brazil, Colombia, Mexico, Guatemala, Ecuador, and Venezuela), where it was introduced through the slave trade. The transmission has been interrupted or eliminated in 11 of 13 foci of the Americas.
As a result of a regional initiative, only about 28,200 people are still in need of continual treatment in Brazil and Venezuela (Yanomami indigenous population). Furthermore, Colombia is the first country in the world to achieve the verification of Onchocerciasis elimination.
- Since 1995, new cases of onchocerciasis blindness have not been reported in the Americas.
- Colombia was the first country of the world receiving the verification of the elimination of onchocerciasis in 2013, followed by Ecuador in 2014, Mexico in 2015, and Guatemala in 2016.
- The Yanomami area in the Amazon region, shared by Brazil and Venezuela, is regarded as the greatest challenge for completing the regional elimination.
- Onchocerciasis is a disease produced by the parasite Onchocerca volvulus, which is transmitted to humans by black flies. This disease also called river blindness because the black flies’ larvae reproduce in rivers and streams with rapid water flow.
- Symptoms include severe itching, disfiguring skin conditions, and visual impairment, including permanent blindness.
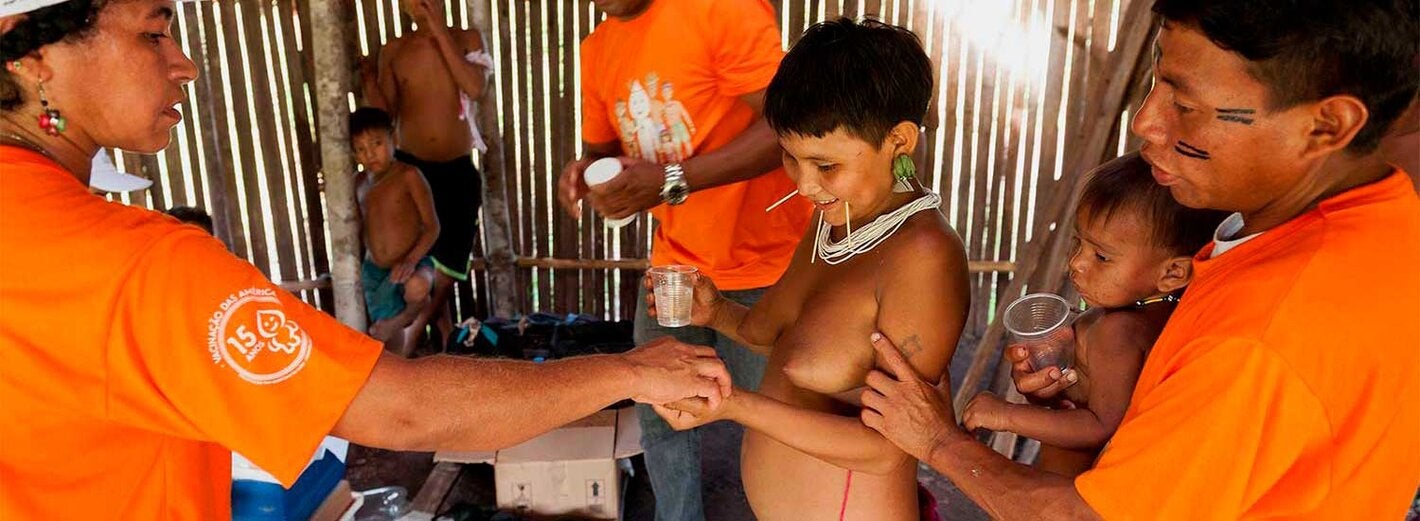
- Globally, it is estimated that there are 18 million people infected and 270,000 blinded by onchocerciasis. It is one of the leading causes of blindness in many African countries (mainly in West Africa and Central Africa), but is also present in Yemen and in Latin America, specifically in Brazil and Venezuela which constitute Yanonami’s area.
- In the Region of the Americas, onchocerciasis transmission has been eliminated in 11 out of the 13 foci. As a result of the regional initiative, only 28,200 people need treatment –at least every 6 months– in the indigenous Yanomami population, living in the Amazon.
- In 1994, the Member States of WHO supported the mass distribution of Ivermectin for onchocerciasis elimination; in addition, call to develop and disseminate the epidemiological methods for onchocerciasis evaluation and/or mapping in endemic countries.
- In 2016, the Directing Council of PAHO/WHO approved resolution CD55.R9, which establishes a plan for the elimination of neglected infectious diseases, including onchocerciasis.
- PAHO/WHO is an active partner of OEPA (Onchocerciasis Elimination Program for the Americas), a regional initiative sponsored by The Carter Center that is in charge of technical operations and coordination of a multinational coalition of agencies, including the six endemic countries in the Americas, Center for Disease Control and Prevention (CDC), United States Agency for International Development (USAID), the Mectizan® Donation Program and other associates whose objectives are to interrupt the transmission, eliminate morbidity of river blindness and eliminate its transmission.