Contents
Preface
Preface by
Dr. Carissa F. Etienne
Chapter I
Responding to
Health Emergencies
Chapter II
Cooperation for Health Development
Chapter III
Institutional Developments
Chapter IV
Conclusions
and Looking Ahead
Preface by
Dr. Carissa F. Etienne
Responding to
Health Emergencies
Cooperation for Health Development
Institutional Developments
Conclusions
and Looking Ahead
.jpeg) To the Member States:
To the Member States:
In accordance with the Constitution of the Pan American Health Organization, I have the honor to present the 2016 annual report on the work of the Pan American Sanitary Bureau, Regional Office for the Americas of the World Health Organization. This report highlights the technical cooperation provided by the Bureau during the period July 2015 through June 2016, within the framework of the 2014-2019 Strategic Plan of the Pan American Health Organization, defined by its Governing Bodies. The report is complemented by the Financial Report of the Director and the Report of the External Auditor for 2015.
Carissa F. Etienne
Director
Pan American Health Organization
The year under review, covering the period of July 2015 to June 2016, was a challenging time for public health in the Americas. Having escaped the potential ravages of the Ebola virus outbreak with only a few imported and secondary infections and two deaths, our Region was once again confronted with another infectious disease emergency in mid-2015. This epidemic was due to the newly emerging Zika virus, whose severe consequences and sequelae would only become gradually apparent over the ensuing months. Following its first detection in Brazil in May 2015, Zika virus had been confirmed in 40 countries and territories across the Region by 21 June 2016.
The rapid spread of the Zika virus was enabled by a confluence of factors. These included the presence of immunologically naïve human populations and the high prevalence of the ubiquitous vector—the Aedes aegypti mosquito—together with increased intraregional travel and mobility. A robust regional public health response to this emergency was mounted, galvanized, and led by the Pan American Sanitary Bureau, which is the Secretariat of the Pan American Health Organization (PAHO).
We provided dynamic leadership and timely technical support and guidance to our Member States as we all grappled with this new challenge. We actively coordinated with the World Health Organization (WHO), other United Nations (UN) agencies, and key partners to ensure value-added joint action. We convened subject matter experts, specialists, and researchers to shape and prioritize a regional research agenda. We stimulated the generation of valuable new knowledge in the face of the numerous questions that arose. Even in the context of limited scientific information, we articulated ethical and evidence-based policy options, especially regarding the associated reproductive health issues.
Indeed, these roles and actions were by no means new or unique for the Bureau. When our predecessor agency, the International Sanitary Bureau, was founded in 1902, its mandate was to provide effective leadership and coordination in health, given the overwhelming communicable disease challenges of that era. This leadership and coordinating role has expanded over the years to include preparedness and response, not only for infectious disease outbreaks but also for disasters and emergencies resulting from natural and manmade hazards. The Zika epidemic, which occupied much of our attention and effort during the 12 months under review, provided an unparalleled opportunity to highlight the Bureau’s role in a health emergency that presented both new and recurrent challenges.
I believe that the results presented in this report will overwhelmingly demonstrate that the policies, procedures, and coordinating mechanisms currently in place at the Bureau and in PAHO Member States are effective for managing public health emergencies in ways that will protect the population, mitigate adverse impacts, and ensure that health services and programs remain functional and effectual during times of greatest need. Our performance in this regard is firmly grounded in a century of experience in coordinating epidemic responses and in more than four decades of experience in disaster preparedness, management, and response.
In addition to the Zika epidemic, the Bureau responded to other health emergencies during the year under review, while continuing to undertake a broad range of non-emergency technical cooperation programs in close collaboration with our Member States. The report details this work and highlights some of the milestones achieved. It also underscores efforts to improve our internal processes and procedures in order to strengthen the efficiency and delivery of technical cooperation, while simultaneously analyzing some of the lessons that we have learned and the challenges that we will face as we continue to implement the PAHO Strategic Plan 2014-2019 and address the global Sustainable Development Goals (SDGs).
In presenting this report to PAHO’s Governing Bodies, I would like to convey my sincerest appreciation to all of our staff for their diligence, dedication, commitment, and arduous work during this 2015–2016 period. I am also extremely grateful to our Member States, donors, and other partners for their invaluable guidance and unstinting support and collaboration over these 12 months. I invite all of PAHO’s stakeholders to join us in further building on the experiences and achievements described in this report in order to fulfill the promise of ensuring the best health possible for every woman, man, and child in our Hemisphere.
 Evolution of an Epidemic Due to the Newly Emerging Zika Virus in the Americas
Evolution of an Epidemic Due to the Newly Emerging Zika Virus in the Americas
The Region of the Americas experienced an explosive epidemic of Zika virus infections during the period of mid-2015 to mid-2016. However, this etiologic agent was first detected in the Western Hemisphere on Chile’s Easter Island in February 2014, coinciding with a series of confirmed Zika outbreaks in French Polynesia, New Caledonia, and other Pacific islands. Chile’s prompt reporting of the presence of Zika virus to the Pan American Sanitary Bureau under the International Health Regulations (IHR) placed the Bureau on alert for the virus’s possible introduction into the continental Americas. At the time, this possibility was not viewed with great alarm, as the known effects of the virus were considered to be relatively mild.
One year later, in February 2015, Brazilian health authorities responded to the Bureau’s request for information on clusters of cases that were presenting with symptoms of fever, muscle and joint pain, rash, and headache in Maranhão, one of the country’s northeastern states. Fourteen of 25 patient samples screened by the Maranhão state laboratory had tested positive for dengue and negative for chikungunya, rubella, and measles. However, this laboratory did not have the capacity to screen for Zika virus. In late April, authorities shared a preliminary report from the state laboratory of Bahia, which noted that samples from Maranhão and other northeastern states had tested positive for Zika. These results were subsequently confirmed by the national reference laboratory in Belém, the Evandro Chagas Institute. On 7 May 2015, PAHO issued its first Epidemiological Alert on Zika virus. In that alert, PAHO described the infection and provided recommendations to Member States for adapting and increasing the sensitivity of their existing dengue and chikungunya surveillance systems in order to detect possible cases of Zika virus infection. The alert also included details on laboratory testing, case management, and prevention and control measures, including recommendations for travelers.
Historically, Zika virus infection had been known to produce mild fever and rash. However, during the 2014 outbreak in French Polynesia, 42 patients had been detected with Guillain-Barré syndrome (GBS), representing a nearly nine-fold increase over the average annual rates. By July 2015 in Brazil’s Bahia State, extensive circulation of Zika virus as well as an increasing incidence in neurological anomalies, including GBS, had been detected.
In August 2015, obstetricians and pediatricians from three other northeastern Brazilian states (Pernambuco, Paraiba, and Rio Grande do Norte) reported what they perceived to be an increase in cases of microcephaly among newborns in their clinical practices. These anecdotal reports were corroborated in October 2015, when a 70-fold increase in detected cases of microcephaly was confirmed in Pernambuco. Many mothers of babies with microcephaly had reported experiencing a febrile rash during their pregnancies.
 In response to these findings, in November 2015 the Bureau deployed a team of experts to Brazil under the auspices of the Global Outbreak Alert and Response Network (GOARN) to assist national and state authorities in characterizing the event. This mission concluded that the increase in microcephaly did not appear to be artifactual or the result of surveillance bias. Based on clinical findings and testing, the team concluded that the microcephaly appeared to be caused by a distinct and novel entity whose impact could be substantial in both the immediate and long term. By the end of December 2015, 11 countries and territories in the Americas were reporting local circulation of Zika virus, from Brazil in the south to Puerto Rico in the north.
In response to these findings, in November 2015 the Bureau deployed a team of experts to Brazil under the auspices of the Global Outbreak Alert and Response Network (GOARN) to assist national and state authorities in characterizing the event. This mission concluded that the increase in microcephaly did not appear to be artifactual or the result of surveillance bias. Based on clinical findings and testing, the team concluded that the microcephaly appeared to be caused by a distinct and novel entity whose impact could be substantial in both the immediate and long term. By the end of December 2015, 11 countries and territories in the Americas were reporting local circulation of Zika virus, from Brazil in the south to Puerto Rico in the north.
With mounting evidence of a link between Zika virus infection and severe complications, particularly microcephaly, the WHO convened a meeting of the IHR Emergency Committee in February 2016 for the specific purpose of considering whether the Zika epidemic met the criteria for a Public Health Emergency of International Concern (PHEIC). That committee concluded that the Zika-associated clusters of microcephaly did meet the IHR criteria for declaration of a Public Health Emergency of International Concern. This prompted WHO Director-General Margaret Chan to formally declare a PHEIC and to call for urgent international coordination and collaboration to better understand the full impact of the Zika virus and its associated complications.
The Bureau proactively coordinated and facilitated a regional Zika response from the earliest stages of the epidemic, months in advance of the PHEIC declaration. The PAHO Alert and Response Operations (ARO) team, which manages event-based surveillance under the WHO IHR mandate, was instrumental in detecting unusual clusters of rash and fever in Brazil’s northeast. Following Brazil’s confirmation of Zika virus circulation in May 2015, the ARO team promptly developed and disseminated recommendations to all Member States to prepare for the possible introduction of Zika virus. In that same month, PAHO’s Director expanded the ARO team into a larger Zika event management team, incorporating additional specialists on surveillance, clinical case management, virology, vector control, and risk communication. Following the Brazil-GOARN findings indicating a strong link between Zika virus and microcephaly, the PAHO Director formally activated the Zika Incident Management System (IMS) on 8 December 2015. The work of ARO and the IMS team in collecting, collating, analyzing, confirming, and communicating information notified by Member States was key to supporting countries as the Zika virus spread rapidly through the Region.
Following the activation of the IMS, the Bureau immediately accessed funds from the PAHO Epidemic Emergency Fund, thereby enabling rapid mobilization of additional human and financial resources to coordinate the regional Zika response. Key actions included the procurement of materials and supplies, including immunoglobulin for treating patients with GBS and insecticides preapproved under the WHO Pesticide Evaluation Scheme (WHOPES). The Bureau mobilized 53 missions to 25 Member States and territories. These missions usually consisted of experts such as neurologists, neonatologists, obstetricians, epidemiologists, virologists, vector control experts, and specialists in research and health services organization. Additionally, the Bureau mounted numerous capacity-building initiatives and other in-country activities in order to provide training to national staff on all aspects critical for an effective response. These capacity-building exercises covered subject areas ranging from laboratory diagnosis and clinical management to vector control and risk communication. In coordination with WHO, Bureau staff drafted or updated 13 technical guidance documents, which advised Member States on the different components of the response. A series of regional and subregional workshops, expert consultations, and other, similar events were also organized. Among them was the first global discussion of a Zika virus research agenda, which brought together representatives from partner organizations that included the U.S. Centers for Disease Control and Prevention (CDC), the Pasteur Institute International Network, and the Oswaldo Cruz Foundation (FIOCRUZ).
 The serious threat posed by Zika virus to pregnant women and their unborn babies, in
combination with the limited scientific knowledge about the virus, its pathogenesis, and resulting
effects, raised difficult ethical issues for health authorities and health care providers. In order to
provide much-needed guidance in this area, the Bureau in April 2016 convened an international
consultation that brought together ethicists and other professionals from ministries of health,
PAHO, and WHO who were involved in the Zika response.
The serious threat posed by Zika virus to pregnant women and their unborn babies, in
combination with the limited scientific knowledge about the virus, its pathogenesis, and resulting
effects, raised difficult ethical issues for health authorities and health care providers. In order to
provide much-needed guidance in this area, the Bureau in April 2016 convened an international
consultation that brought together ethicists and other professionals from ministries of health,
PAHO, and WHO who were involved in the Zika response.
The following recommendations emerged from that consultation:
 As evidence of a link between Zika virus infection and birth defects mounted, the Bureau in January 2016 convened internal and external experts on maternal and perinatal health, genetics, epidemiology, surveillance, and pediatric neurology to develop guidelines for both the diagnosis and surveillance of microcephaly (eventually considered as part of the more broadly defined congenital Zika syndrome, or CZS) as well as for the care of pregnant women exposed to Zika virus and newborns with microcephaly/CZS. Technical missions with expertise in the care of pregnant women were deployed to Bolivia, Colombia, the Dominican Republic, Ecuador, El Salvador, Haiti, Honduras, Guatemala, and Panama. Bureau experts coordinated with WHO colleagues for the purpose of developing criteria for early diagnosis of microcephaly/CZS, utilizing ultrasound.
As evidence of a link between Zika virus infection and birth defects mounted, the Bureau in January 2016 convened internal and external experts on maternal and perinatal health, genetics, epidemiology, surveillance, and pediatric neurology to develop guidelines for both the diagnosis and surveillance of microcephaly (eventually considered as part of the more broadly defined congenital Zika syndrome, or CZS) as well as for the care of pregnant women exposed to Zika virus and newborns with microcephaly/CZS. Technical missions with expertise in the care of pregnant women were deployed to Bolivia, Colombia, the Dominican Republic, Ecuador, El Salvador, Haiti, Honduras, Guatemala, and Panama. Bureau experts coordinated with WHO colleagues for the purpose of developing criteria for early diagnosis of microcephaly/CZS, utilizing ultrasound.
The Bureau developed guidelines for psychosocial support of pregnant women in areas with Zika virus circulation and organized three webinars to help disseminate that information. Other guidelines were produced on issues that included safe blood transfusions and the production of safe blood products within the context of this epidemic.
In addition to widely disseminating written technical guidance, the Bureau provided frequent updates on the Zika epidemic for key stakeholders in Member States as well as for the Permanent Council of the Organization of American States (OAS); the Members and Associate States of MERCOSUR; the ministers of health of the Union of South American Nations (UNASUR); and the members of the United Nations Development Group-LAC (UNDG-LAC). Briefings on Zika and its impact on women and babies were provided for the Inter-American Commission of Women, the Regional Task Force for the Reduction of Maternal Mortality (GTR), and the International Economic Forum on Latin America and the Caribbean (LACFORUM). The Director used the opportunities provided by her visits to Member States to update national audiences on the evolving Zika situation and the Bureau’s multifaceted response, such as through town hall meetings and other events in Colombia, Cuba, Ecuador, Grenada, and Saint Vincent and the Grenadines.
Building on previous technical cooperation initiatives for health systems strengthening and Ebola preparedness, the Bureau developed a new assessment instrument for evaluating countries’ capacities to respond to the Zika epidemic and complications of the disease. Applied in countries that included Bolivia, Colombia, Costa Rica, the Dominican Republic, Haiti, Honduras, Paraguay, and Suriname, the instrument helped national health authorities to identify areas for strengthening. In a separate effort, the Bureau partnered with the World Bank (WB) and the Inter-American Development Bank (IDB) to develop a combined assessment and costing tool to identify gaps in national health systems’ response capacities and to estimate the costs of needed interventions. Joint missions of experts from the three institutions (PAHO, WB, IDB) were deployed to Dominica and Honduras to pilot test this new instrument. It has since been applied in El Salvador, Grenada, Guyana, Haiti, Nicaragua, Panama, and two states in Brazil.
The Bureau also collaborated with WHO and the United Nations Children’s Fund (UNICEF) to develop a target product profile for a Zika diagnostic test that could readily be utilized in national health services.
 To address the critical issue of vector control, PAHO’s Director constituted a new Technical Advisory Group in Public Health Entomology, which met for the first time in March 2016. Participants included experts from WHO, FIOCRUZ, U.S. CDC, the United States Agency for International Development (USAID), Australia’s Monash University, the Bill and Melinda Gates Foundation, and the U.S. White House Office of Science and Technology Policy. The meeting recommendations included enhancing mosquito control and surveillance and increasing training in these areas; developing a new protocol for monitoring insecticide resistance; and evaluating the impact and cost-effectiveness of current vector control methods as well as new and supplemental tools, such as genetically modified and/or sterile mosquito technologies. The group also emphasized the need for stronger intersectoral action, especially through partnerships with communities and with relevant productive sectors to develop, implement, and sustain effective and economically viable actions to reduce mosquito populations.
To address the critical issue of vector control, PAHO’s Director constituted a new Technical Advisory Group in Public Health Entomology, which met for the first time in March 2016. Participants included experts from WHO, FIOCRUZ, U.S. CDC, the United States Agency for International Development (USAID), Australia’s Monash University, the Bill and Melinda Gates Foundation, and the U.S. White House Office of Science and Technology Policy. The meeting recommendations included enhancing mosquito control and surveillance and increasing training in these areas; developing a new protocol for monitoring insecticide resistance; and evaluating the impact and cost-effectiveness of current vector control methods as well as new and supplemental tools, such as genetically modified and/or sterile mosquito technologies. The group also emphasized the need for stronger intersectoral action, especially through partnerships with communities and with relevant productive sectors to develop, implement, and sustain effective and economically viable actions to reduce mosquito populations.
Amid increasing public concern about the risks posed by the Zika outbreak in Brazil to athletes and fans attending the upcoming summer Olympics in Rio de Janeiro, the Bureau provided information and analysis for consideration at the June 2016 meeting of the IHR Emergency Committee. The Bureau’s report, together with information provided by Brazil, reviewed recent epidemiological trends and demonstrated a decline in the current incidence of Zika infections. It also noted that the transmission of dengue virus, which is spread by the same Aedes aegypti vector, has been historically low during the winter months of August and September in Brazil. The IHR Emergency Committee concluded that the risk of further international spread of Zika as a result of the Olympic and Paralympic Games was very low, and that the Committee’s earlier advice that there should be no general restrictions on travel and trade remained valid.
The report provided by the Bureau to the IHR Emergency Committee incorporated information gathered during a series of missions to Brazil in which the Bureau’s Zika IMS team advised national health authorities on preparations for the Olympic Games in areas that included information management, alert and response triggering, command and control across key levels of government (municipal, state, and federal), and transparency and sharing of health information with other PAHO Member States and stakeholders.
Under an agreement with the International Olympic Committee and in partnership with Brazilian authorities, the Bureau and WHO provided support for reducing the risks of Zika infections among Olympic athletes and visitors as well as for cutting the risks of water-borne illness among athletes competing in Olympic water sports.
To prevent Zika infections, the Bureau and WHO recommended several actions. One was fumigation and indoor spraying to decrease adult mosquito populations. Another was eliminating potential breeding sites in and around Olympic sports venues, lodgings for athletes, and hotels. A third was community engagement to eliminate potential breeding sites in nearby homes, schools, and workplaces. The Bureau and WHO also recommended distribution of insect repellents and health promotion materials to visitors and athletes. The Bureau and WHO also suggested systematic outreach by Brazilian health authorities to Olympic delegations before and during the games, to inform them about the measures being implemented and any changes in the epidemiological situation.
To help prevent water-borne illness, Bureau and WHO staff helped Brazilian authorities develop a methodology for monitoring the quality of recreational waters through bacteriological analysis, and proposed actions to reduce contamination. A Bureau-organized workshop in Brazil in November 2015 produced additional recommendations for strengthening social communication, public information, and health promotion. In March 2016, a Bureau mission visited the water test sites and reviewed the water quality criteria being used to monitor those sites. As of mid-June 2016, the Bureau continued to support Brazil’s efforts in this area.
 In supporting Brazil and providing regional coordination for the Zika response, the Bureau drew heavily on existing partnerships and networks, such as the Dengue Laboratory Network of the Americas (RELDA), whose participating laboratories include the National Institute of Human Viral Diseases (INEVH) in Argentina; the Evandro Chagas Institute and FIOCRUZ in Brazil; Cuba’s Pedro Kouri Institute; the Pasteur Institute in French Guiana; Mexico’s National Institute of Epidemiologic Diagnosis and Reference; Panama’s Gorgas Memorial Institute for Health Studies; and the U.S. CDC in Puerto Rico. These laboratories agreed to expand the scope of the network in order to provide integrated laboratory surveillance of all arboviruses.
In supporting Brazil and providing regional coordination for the Zika response, the Bureau drew heavily on existing partnerships and networks, such as the Dengue Laboratory Network of the Americas (RELDA), whose participating laboratories include the National Institute of Human Viral Diseases (INEVH) in Argentina; the Evandro Chagas Institute and FIOCRUZ in Brazil; Cuba’s Pedro Kouri Institute; the Pasteur Institute in French Guiana; Mexico’s National Institute of Epidemiologic Diagnosis and Reference; Panama’s Gorgas Memorial Institute for Health Studies; and the U.S. CDC in Puerto Rico. These laboratories agreed to expand the scope of the network in order to provide integrated laboratory surveillance of all arboviruses.
Other crucial support for the Bureau’s Zika response came from the WHO Contingency Fund for Emergencies (CFE), the Public Health Agency of Canada, Global Affairs Canada, USAID, the Bill and Melinda Gates Foundation, and Norway (through WHO’s CFE).
Many aspects of this Zika epidemic have reinforced some of the Bureau’s ongoing technical cooperation priorities and approaches, while simultaneously suggesting the need for changing emphases in other areas. This is certainly the case regarding vector control and management. The current Zika epidemic, the large and recurrent outbreaks of dengue fever over the past three decades, and the recent emergence and rapid spread of chikungunya virus have underscored the magnitude of the Aedes aegypti infestation in the Region and have also exposed the failure of countries to control this vector of major public health importance. Although the specific lessons learned for vector control are not new, the Zika epidemic has provided added impetus for their urgent application. A central lesson is that mosquito populations and human exposure are associated with many environmental and social determinants of health. Therefore, vector control programs must be comprehensive, integrated, intersectoral, and participatory. Another key lesson is that resources devoted to vector control efforts must be sustained at appropriate levels even when such efforts lose high visibility and attention because of their own success. At the same time, innovation is essential for developing new and more effective ways of controlling mosquito populations.
A related lesson learned is the need for integrated surveillance, prevention, and control of arboviral diseases of public health importance. This is particularly true for dengue, chikungunya, yellow fever, and Zika, which are all transmitted by the same mosquito vector. These integrated efforts should include entomological surveillance of mosquito species, vector monitoring for insecticide resistance, and laboratory surveillance for viruses present in vectors.
The Region’s Zika experience has also demonstrated the critical need for frontline health providers to maintain a high index of suspicion and to be alert to atypical clinical presentations and events, in order to ensure the timely detection of an emerging disease. On the regional level, the same judgment and awareness are required when reviewing data generated by event-based surveillance and when responding appropriately even in the absence of strong confirmatory evidence. The Bureau’s guiding principle in responding to Zika has been to act with caution but with commensurate urgency, relying on the best evidence available. In this context, well-tailored risk communication activities are essential for truthfully and transparently conveying uncertainties in a manner that merits and preserves the public’s trust.
Previous work initiated by the Bureau and PAHO Member States to strengthen epidemic alert and response capacity and the role of IHR national focal points in countries contributed positively to the Zika regional response. Originally undertaken to advance national and regional compliance with the IHR, these efforts had been accelerated during the 2014 preparations for the possible introduction of Ebola virus. These exercises had provided many countries with the opportunity to identify and address gaps in their epidemic preparedness. In addition, the procedures and channels established as part of IHR proved effective and indeed essential for exchanging and processing outbreak information to guide the national, regional, and global responses to Zika’s spread.
 Within the Bureau, responding to the Zika epidemic has galvanized intense inter-programmatic collaboration and has produced new synergies between technical programs, including ones that deal with communicable disease surveillance and control, disaster preparedness and response, health systems and services, and women’s and children’s health, among others.
Within the Bureau, responding to the Zika epidemic has galvanized intense inter-programmatic collaboration and has produced new synergies between technical programs, including ones that deal with communicable disease surveillance and control, disaster preparedness and response, health systems and services, and women’s and children’s health, among others.
Like chikungunya before it, Zika has highlighted persisting weaknesses and gaps in the capacity of national vector control programs and health systems to adapt to new threats. In this context, the epidemic has reinforced the overarching emphasis that the Bureau has placed on building strong, resilient health systems and advancing universal access and coverage. It has also highlighted other technical cooperation priorities such as achieving full compliance with the IHR and integrating surveillance, prevention, and control of arboviral diseases of public health importance.
At this time, much remains to be learned about Zika. However, the proactive regional response guided by visionary public health leadership and coordination during the review period has led to a much better understanding of the spectrum of disease caused by this virus. As of 30 June 2016, Zika-associated neurological disorders, including GBS, had been reported in 15 countries and territories in the Americas. Microcephaly related to locally acquired Zika infections had been reported in 7 locations: Brazil, Colombia, El Salvador, French Guiana, Martinique, Panama, and Puerto Rico. In addition to the strong temporal and spatial associations identified through surveillance data analyses, other evidence of a link between Zika and these complications included: a) the identification of Zika virus in brain tissue of affected fetuses and newborns; b) the documentation of the infectivity of Zika virus in neural stem cells; c) the identification of specific cell receptors that are favored by the virus; and d) the detection of Zika infection during specific phases of prenatal development that were consistent with observed fetal defects. The critical finding that Zika virus can also be transmitted sexually gives great cause for concern, as this transmission route has the potential to greatly amplify the spread of this virus even in the absence of competent mosquito vectors.
There are approximately 500 million persons living in areas at risk for the vector transmission of Zika virus in Latin America and the Caribbean. The impact of this virus has already proved to be substantial. The virus is likely to place a heavy burden on health services (including mental health services), especially in the treatment of serious complications, and on society, particularly for women. Zika will remain on the regional public health agenda for 2016–2017 and beyond, and PAHO will continue to respond appropriately and effectively.
 In addition to its Zika-related work, the Bureau provided key support to Member States for several other important health emergencies during the period under review. The most serious of these resulted from a 7.8-magnitude earthquake that struck Ecuador on 16 April 2016, affecting over 720,000 people. The disaster resulted in the loss of 663 lives and forced more than 33,000 persons to seek refuge in shelters. Thirty-nine health centers and hospitals were damaged, and 20 were unable to continue functioning.
In addition to its Zika-related work, the Bureau provided key support to Member States for several other important health emergencies during the period under review. The most serious of these resulted from a 7.8-magnitude earthquake that struck Ecuador on 16 April 2016, affecting over 720,000 people. The disaster resulted in the loss of 663 lives and forced more than 33,000 persons to seek refuge in shelters. Thirty-nine health centers and hospitals were damaged, and 20 were unable to continue functioning.
In response, the Bureau activated its Institutional Emergency Response framework and emergency operations centers (EOCs) at both the Headquarters and country office levels. The latter immediately mobilized staff and resources to support Ecuador’s health authorities. Shortly afterwards, technical staff from Headquarters and other country offices were deployed to support coordination of the humanitarian response (including emergency medical teams), damage and needs assessments (particularly for health infrastructure), information management and communication, and logistical supply management. Bureau staff also worked closely with national authorities on epidemiological surveillance, the restoration of health services, immunization, and mental health.
With the Bureau’s support, health authorities set up two staging areas, in Quito and Manta, to register and coordinate the movement of medicines and medical supplies into and out of warehouses, using the Bureau’s Logistics Support System/Humanitarian Supply Management System (LSS/SUMA).
 For the first time in the Region, Ecuador’s Ministry of Public Health utilized the minimum standards and coordinating mechanisms that had been promoted by PAHO/WHO for the deployment of national and international emergency medical teams (EMTs) (see box below). An Emergency Medical Teams Information and Medical Coordination Cell (CICOM) was established in Quito’s ECU 911 Emergency Center under the responsibility of the Ministry of Public Health’s International Cooperation Department. CICOM facilitated the registration and deployment of EMTs to disaster-affected areas, assisted with the prioritization of the work of health responders, and improved the quality of information regarding health coverage. The EMTs that were deployed consisted of more than 20 national and 5 international teams, reflecting the success of the Bureau’s ongoing technical efforts and support aimed at enhancing national capacity to respond to emergencies.
For the first time in the Region, Ecuador’s Ministry of Public Health utilized the minimum standards and coordinating mechanisms that had been promoted by PAHO/WHO for the deployment of national and international emergency medical teams (EMTs) (see box below). An Emergency Medical Teams Information and Medical Coordination Cell (CICOM) was established in Quito’s ECU 911 Emergency Center under the responsibility of the Ministry of Public Health’s International Cooperation Department. CICOM facilitated the registration and deployment of EMTs to disaster-affected areas, assisted with the prioritization of the work of health responders, and improved the quality of information regarding health coverage. The EMTs that were deployed consisted of more than 20 national and 5 international teams, reflecting the success of the Bureau’s ongoing technical efforts and support aimed at enhancing national capacity to respond to emergencies.
In coordination with the UN Office for the Coordination of Humanitarian Affairs (OCHA), Bureau staff led the development of the health sector component of an urgent appeal to raise funds for humanitarian assistance. Launched on 22 April 2016, the appeal included a US$ 4.5 million request from the health sector to address the health needs of 110,000 people for three months. The goal was to increase access to essential health services, including emergency care, maternal and child care, and nutrition services, and to reduce health risks through surveillance, access to reproductive health services, and services for victims of gender-based violence. Bureau staff also supported the development and launch of an urgent appeal on behalf of the water, sanitation, and hygiene sector in Ecuador. By the end of May 2016, the Bureau had received funds or commitments of more than US$ 1.5 million (from Canada, Italy, the United States, and the UN Central Emergency Response Fund (CERF)) to support the Bureau’s technical cooperation in this area.
 The Bureau has been supporting the rapid mobilization of skilled emergency medical teams (EMTs) to Member States affected by sudden-onset disasters or outbreaks, under principles and procedures laid out in the 2014 PAHO Plan of Action for the Coordination of Humanitarian Assistance (CD53/12 [2014]). These include globally agreed minimum standards and requirements for staffing and equipping EMTs; procedures for requesting, accepting, and receiving EMTs; steps for coordinating the integration of EMTs with health emergency operations center (EOCs); and guidance for streamlining immigration, customs, and logistics procedures to facilitate EMT deployments. The PAHO Plan of Action also includes guidance on forming national-level EMTs and developing mechanisms to register both national and international teams.
The Bureau has been supporting the rapid mobilization of skilled emergency medical teams (EMTs) to Member States affected by sudden-onset disasters or outbreaks, under principles and procedures laid out in the 2014 PAHO Plan of Action for the Coordination of Humanitarian Assistance (CD53/12 [2014]). These include globally agreed minimum standards and requirements for staffing and equipping EMTs; procedures for requesting, accepting, and receiving EMTs; steps for coordinating the integration of EMTs with health emergency operations center (EOCs); and guidance for streamlining immigration, customs, and logistics procedures to facilitate EMT deployments. The PAHO Plan of Action also includes guidance on forming national-level EMTs and developing mechanisms to register both national and international teams.
During 2015-2016, the Bureau focused major efforts on building country capacity for coordinating EMTs. National workshops were convened in Chile, Colombia, Costa Rica, Ecuador, Guatemala, and Peru. They provided training for 183 participants on the procedures and tools for requesting, receiving, and deploying EMTs and for improving the coordination of EMTs through health EOCs and the establishment of EMT information cells (CICOM).
In December 2015, the Bureau partnered with WHO’s Emergency Medical Teams Secretariat to host a global EMT meeting. Participants included 150 representatives of governments (from more than 30 WHO Member States), civil society, the private sector, and international organizations. Among issues discussed were strengthening global collaboration and capacity, the EMT quality assurance framework, national mechanisms for team registration and coordination, and minimum standards for national and international teams.
The meeting report is available at: http://www.paho.org/disasters/index.php?option=com_docman&task=doc_download&gid=2409&Itemid=&lang=en.
 In early October 2015, the Bahamas was struck by Hurricane Joaquin, a category-4 storm that heavily impacted six islands, with more than 14,000 inhabitants. High winds, high tides, flooding, and debris from heavy rains restricted access by air, road, and sea, while downed power lines and damaged generators interrupted electricity supplies, disrupting the cold chain in several health centers.
In early October 2015, the Bahamas was struck by Hurricane Joaquin, a category-4 storm that heavily impacted six islands, with more than 14,000 inhabitants. High winds, high tides, flooding, and debris from heavy rains restricted access by air, road, and sea, while downed power lines and damaged generators interrupted electricity supplies, disrupting the cold chain in several health centers.
As the only United Nations agency resident in the Bahamas, the PAHO Country Office was in a unique position to assist the Government from the onset of the disaster. In the days following Hurricane Joaquin, the Bureau deployed experts in disaster management from its Regional Disaster Response Team to assist the Ministry of Health and the Ministry of the Environment with an assessment of the hurricane’s impact and in identifying priority interventions, while also supporting supply management and coordination efforts.
In addition, the Bureau donated more than US$ 100,000 worth of essential supplies to facilitate communication on the ground, ensure continued health care delivery, and support response interventions in the areas of water and sanitation and vector control. These supplies included generators, manual diesel pumps, and diesel storage drums to restore the functioning of several affected health care facilities. Essential medicines and vaccines were purchased to replace damaged and lost stocks, along with refrigeration units to restore the cold chain. Other Bureau contributions included Iridium satellite telephones to support communication among health facilities and insecticide and foggers to allow the Department of Environmental Health Services to intensify its vector-control interventions.
The joint response by the Bureau and the Bahamian authorities played a critical role in improving the quality of life and health of the population impacted by Hurricane Joaquin.
 Building on PAHO’s Safe Hospitals Initiative, national stakeholders in the Caribbean are rallying around a new effort to make hospitals “smart.” This Bureau-led approach represents a transition away from the traditional disaster response model, by proactively incorporating measures for climate adaptation, mitigation, and preparedness to reduce the impact of disasters on health. This approach simultaneously seeks to reduce the environmental footprint of the health sector, one of the heaviest consumers of energy. Health care facilities are “smart” when they link their structural and operational safety with green interventions, at a reasonable cost-to-benefit ratio.
Building on PAHO’s Safe Hospitals Initiative, national stakeholders in the Caribbean are rallying around a new effort to make hospitals “smart.” This Bureau-led approach represents a transition away from the traditional disaster response model, by proactively incorporating measures for climate adaptation, mitigation, and preparedness to reduce the impact of disasters on health. This approach simultaneously seeks to reduce the environmental footprint of the health sector, one of the heaviest consumers of energy. Health care facilities are “smart” when they link their structural and operational safety with green interventions, at a reasonable cost-to-benefit ratio.
In May 2015, the Bureau secured commitments from the Governments of Dominica, Grenada, Saint Lucia, and Saint Vincent and the Grenadines, along with funding of £8.3 million (about US$ 11 million) from the U.K. Department for International Development (DFID), to make several health facilities in each country “smart.” Recognizing the value of a platform that links disaster risk reduction and climate change, the Prime Minister of the United Kingdom in September 2015 announced additional funding of £30 million (nearly US$ 40 million) to extend the project to Belize, Guyana, and Jamaica and to increase support for the original participating countries. The project’s goal is to make 50 health facilities in these countries safer and greener by 2020. In addition, the project will assess a total of 600 facilities to document, in an online database, their needs for future improvements. By June 2015, all 173 facilities in the initial four project countries had been assessed, and 14 had been identified as priority facilities for retrofitting. In addition, 62 national professionals had received training on applying the SMART toolkit and managing retrofitting works.
The bulk of the project’s funding will support renovations to ensure that health facilities are better able to withstand natural disasters and climate variability. This work includes the strengthening of roofs and structures; the installation of hurricane shutters and energy and water conservation devices; and improvements in storm drainage, access, and fire safety. The Bureau is providing technical support to ensure that appropriate disaster response plans and equipment are also in place for each facility and that maintenance and conservation procedures are established and utilized.
The Bureau also supported the rollout of a public relations strategy and a knowledge, attitudes, and practices (KAP) survey. Those efforts resulted in increased awareness of the project among other agencies and multilateral funding institutions and greater interest on their part in applying the concept to new health facilities, schools, and hotels.
 Tropical Storm Erika, the fifth named storm of the 2015 Atlantic hurricane season, deluged Dominica in late August with 13 inches of rain in a 12-hour period, resulting in severe and widespread flooding and extensive damage to homes, health facilities, and other essential infrastructure, including roads, bridges, and water supply and communication systems. The Bureau rapidly deployed its Regional Disaster Response Team to assist Dominican authorities with emergency coordination, damage assessment, disease surveillance, and vector prevention and control. In the following weeks, the Bureau coordinated medical personnel who had arrived from neighboring countries to help restore health services in affected areas. The Bureau provided financial, logistical, and coordinating support for these efforts and channeled nearly US$ 800,000 in voluntary contributions from the U.K. DFID, the European Civil Protection and Humanitarian Aid Operations (ECHO), and Global Affairs Canada (GAC, formerly DFATD) to support health care delivery, provide access to safe water and sanitation, and reduce environmental health risks. The funds allowed the Bureau to procure essential medicines and health supplies, critical vector-control equipment and supplies, water testing kits and purification tablets, and rainwater harvesting systems, which were installed in 39 health centers.
Tropical Storm Erika, the fifth named storm of the 2015 Atlantic hurricane season, deluged Dominica in late August with 13 inches of rain in a 12-hour period, resulting in severe and widespread flooding and extensive damage to homes, health facilities, and other essential infrastructure, including roads, bridges, and water supply and communication systems. The Bureau rapidly deployed its Regional Disaster Response Team to assist Dominican authorities with emergency coordination, damage assessment, disease surveillance, and vector prevention and control. In the following weeks, the Bureau coordinated medical personnel who had arrived from neighboring countries to help restore health services in affected areas. The Bureau provided financial, logistical, and coordinating support for these efforts and channeled nearly US$ 800,000 in voluntary contributions from the U.K. DFID, the European Civil Protection and Humanitarian Aid Operations (ECHO), and Global Affairs Canada (GAC, formerly DFATD) to support health care delivery, provide access to safe water and sanitation, and reduce environmental health risks. The funds allowed the Bureau to procure essential medicines and health supplies, critical vector-control equipment and supplies, water testing kits and purification tablets, and rainwater harvesting systems, which were installed in 39 health centers.
Thanks to the combined efforts of the Dominican Government, the Bureau, neighboring countries, and other partners, health services were reestablished in 98% of the country’s health facilities within six months, and outbreaks of vector-borne and water-transmitted diseases were successfully contained.
 The El Niño Southern Oscillation (ENSO) brought heavy rains and severe flooding to several South American countries during this review period. Paraguay was one of the most seriously affected countries, with 11 of its 17 departments experiencing severe floods, some 132,000 people displaced, and major crop and food stock losses. Paraguay’s National Emergency Department facilitated the evacuation of over 60,000 persons from the affected areas and activated a large network of shelters to host the displaced populations.
The El Niño Southern Oscillation (ENSO) brought heavy rains and severe flooding to several South American countries during this review period. Paraguay was one of the most seriously affected countries, with 11 of its 17 departments experiencing severe floods, some 132,000 people displaced, and major crop and food stock losses. Paraguay’s National Emergency Department facilitated the evacuation of over 60,000 persons from the affected areas and activated a large network of shelters to host the displaced populations.
The Bureau assisted national and local authorities with rapid needs assessments (including of damaged health facilities), information management, emergency response planning, and overall coordination of the health sector response. The Bureau also procured communication equipment for the emergency situation rooms of the Ministry of Health and the National Emergency Secretariat and provided tents for medical field consultations. To reduce the risk of water contamination and water-borne illness, the Bureau helped with the construction of two Imhoff sewage tanks and the rehabilitation of 60 portable sanitary modules that ensured proper sanitation and hygiene in shelters.
With the return of the rainy season in May 2016, the Bureau continued to monitor Paraguay’s emergency situation, which remained critical as of June 2016, with rivers at above-normal water levels making it unsafe for displaced persons to return to their homes.
El Niño also affected Guatemala, but its impact was exacerbated by a prolonged dry spell that began in late 2013 and lasted through March 2016. These combined weather phenomena produced major crop losses, an ensuing food crisis, and higher rates of acute malnutrition in the most vulnerable groups: children under 5 and pregnant women. As of early 2016, an estimated 248,000 families were in need of humanitarian assistance.
The Bureau channeled over US$ 1.1 million from CERF and ECHO to implement lifesaving interventions and preventive actions to address the health and nutritional needs of children under 5 years of age, pregnant women, and women of childbearing age suffering from or at risk of acute malnutrition. The Bureau also supported the coordination and preparedness efforts of the Ministry of Health and other partners.
Specific Bureau support included training health workers on detection and treatment protocols for moderate and severe acute malnutrition, acute respiratory infections, and foodborne diseases; establishing mobile health teams; procuring essential health supplies; monitoring and reporting health data; implementing immunization campaigns; and putting in place protective measures for pregnant and lactating women.
 The Bureau provided critical support to several Member States that faced unexpected migrant flows during the reporting period. These outreach activities were extended to Venezuelan migrants in Colombia, Cuban migrants in Central America, and residents of the Dominican Republic of Haitian descent who were repatriated to Haiti.
The Bureau provided critical support to several Member States that faced unexpected migrant flows during the reporting period. These outreach activities were extended to Venezuelan migrants in Colombia, Cuban migrants in Central America, and residents of the Dominican Republic of Haitian descent who were repatriated to Haiti.
During 2015 and 2016, Colombia received a massive influx of both Venezuelan immigrants and returning Colombian expatriates who had been living in Venezuela. Between October 2015 and May 2016, some 171,000 migrants crossed the border, and as of June 2016 roughly 2,000 migrants were being registered each week in the department of Norte de Santander, along the Venezuelan border. This situation had a major impact on Colombia’s border communities. It placed excessive demands on public services that were already limited, in an area that was relatively isolated from the rest of the country.
As lead of the Country Health Cluster in Colombia, the Bureau facilitated the mapping of actors and humanitarian interventions conducted in the border areas and supported the development of a response plan, under the coordination of the United Nations Office of the High Commissioner for Refugees (UNHCR) and OCHA.
The Bureau also assisted the national health authorities with identifying and monitoring health risks and needs. One special concern was the need for basic health services and sanitation in shelters. Another issue was potential changes in the population’s health profile as a result of low vaccination rates among the Venezuelan migrants. Bureau staff worked collaboratively with health authorities to increase vaccination coverage and expand resources for health care, while also strengthening prevention and control efforts for Zika, malaria, and other infectious diseases circulating in those areas.
In late 2015, thousands of Cuban nationals left their home country and traveled through Ecuador (which had temporarily waived its visa requirements), apparently intending to continue northward towards the United States. Many found themselves stranded in Costa Rica, however, after Nicaragua closed its border. When Costa Rica began issuing a more limited number of transit visas, newer Cuban migrants began congregating in northern Panama. By mid-December, more than 4,200 migrants were being housed in 31 shelters in Costa Rica’s northern border districts. An additional 2,200 were awaiting visas to cross from Costa Rica into Nicaragua or into Costa Rica from Panama.
At the request of the Costa Rican Government, the Bureau mobilized support for the coordination and response efforts of the Ministry of Health and the National Emergency Commission. In collaboration with other UN agencies and international humanitarian actors, the Bureau helped activate a Coordination Center for Health Operations and deployed staff from both the Country Office in San Jose and the Regional Disaster Response Team to equip and manage shelters, conduct needs assessments, evaluate health risks, and support the Ministry of Health’s efforts to provide psychosocial support to the migrants.
Hispaniola narrowly averted a potential health crisis when an estimated 100,000 Haitians who had been living in the Dominican Republic were deported or returned voluntarily to Haiti following a legal ruling that retroactively rescinded the birthright citizenship of Dominicans of Haitian descent.
The beginning of the mass repatriation, in June 2015, coincided with the start of the hurricane season as well as with legislative, municipal, and presidential elections in Haiti. These factors, combined with diminished operational and financial capacities of the Humanitarian Country Team in Haiti, raised fears of a potential humanitarian crisis. A major health concern was an increased risk of cholera outbreaks among deported migrants in border areas.
In response, the Bureau, with financial support from Canada, partnered with Haiti’s Ministry of Public Health and Population (MSPP) to establish an advance health post in the commune of Ouanaminthe, in the Northeast Department, and helped reinforce existing border health centers to ensure the availability of health care for returnees. The Bureau and the MSPP enhanced epidemiological surveillance, especially in border departments, while strengthening clinical capacity for treating and managing cases of diarrheal illness. Other Bureau support included procurement of emergency health kits to cover the basic health needs of 10,000 patients for three months, and supplemental kits with postexposure prophylaxis and treatment for malaria.
As a result of these and other actions undertaken by the MSPP, the Bureau, other humanitarian partners that included OCHA, the International Organization for Migration (IOM), and local and international nongovernmental organizations, no cholera outbreak was directly linked to the arrival of deported Haitians in border communes. Incidence rates of diarrheal diseases and acute respiratory infections also remained stable in these areas. As of June 2016, more returnees continued to cross the border into Haiti.
On 14 October 2015, a fire began in a storage depot that contained old electrical transformers filled with polychlorinated biphenyls (PCBs) in San Lorenzo, Paraguay. An estimated 4,000 people residing in the immediate vicinity were exposed to the fumes, which were visible from 5 kilometers away. The fumes of incomplete combustion of PCBs are a major source of dioxins and furans that have been classified as Group 1 carcinogens by the International Agency for Research on Cancer (IARC). The incident in Paraguay was considered a major event, one of the largest involving the risk of exposure to dioxins in recent history.
In response to a request from the Government of Paraguay, the Bureau provided technical guidance to the Ministry of Health for immediate response and preliminary public health risk assessment, as part of a joint multiagency team that also included staff of the UN Environmental Program (UNEP), the UN Development Program (UNDP), and OCHA, as well as from the secretariats of the Basel, Rotterdam, and Stockholm Environmental Conventions and Paraguayan Government agencies.
The Bureau delivered a rapid assessment report, which indicated a high possibility of exposure to dioxins. Based on extensive interviews and more than 400 blood samples, 252 people were found to have signs and symptoms that could be directly related to the incident. These persons were referred for medical attention and follow-up by the country’s health services.
Recognizing that long-term effects of dioxin exposures, such as cancers or problems related to endocrine disruption, may take decades to develop, the Bureau recommended a series of follow-up actions, which were incorporated into a proposal for funding from the United Nations Industrial Development Organization (UNIDO).
The interagency and intersectoral collaboration in responding to this event led to a proposal by the Inter-American Development Bank (IDB) in collaboration with the UNDP and others to establish a permanent roundtable involving all involved agencies (including PAHO) and sectors to coordinate efforts to strengthen Paraguay’s capacity to prevent, prepare for, and respond to technological emergencies and incidents.
 Along with the Zika epidemic, other health emergencies that occurred during this review period provided new evidence of the critical importance of the Bureau’s technical cooperation with its Member States in epidemic and emergency preparedness, IHR core capacity-building, and disaster risk reduction. In addition, however, these emergencies revealed persisting vulnerabilities and fragilities in national health systems throughout the Americas. As was demonstrated so clearly and poignantly by the Ebola epidemic in West Africa, fragile health systems are not able to effectively cope with serious disease outbreaks, nor can they efficiently respond to natural and other types of disasters or sustained stresses such as economic downturns or poor stewardship.
Along with the Zika epidemic, other health emergencies that occurred during this review period provided new evidence of the critical importance of the Bureau’s technical cooperation with its Member States in epidemic and emergency preparedness, IHR core capacity-building, and disaster risk reduction. In addition, however, these emergencies revealed persisting vulnerabilities and fragilities in national health systems throughout the Americas. As was demonstrated so clearly and poignantly by the Ebola epidemic in West Africa, fragile health systems are not able to effectively cope with serious disease outbreaks, nor can they efficiently respond to natural and other types of disasters or sustained stresses such as economic downturns or poor stewardship.
Building on its technical cooperation in the areas of health systems strengthening and universal health, the Bureau developed a policy document on resilient health systems (CD55/9 [2016]), including the rationale and lines of action for this effort. The policy affirms that investing in health systems resilience is considerably more cost-efficient than financing emergency responses.
Key areas of investment for health system resilience include strengthening of the
essential public health functions (especially governance and regulation), health
surveillance and health information systems, and risk reduction and communication.
Investments in primary health care services are also crucial and should be aimed at
ensuring that these services are universally available and are part of networks that are
adaptable and responsive.
 A key component of these efforts is related to the IHR. To build resilience, the
implementation of the IHR should be approached as a holistic process and should also be
embedded in national policy development and planning, legislative action, and regulatory
frameworks. The implementation should include improvements in health services
organization to support infection prevention and control, strengthening of health
surveillance networks and laboratory capacity, and development of health workforce
competencies in the areas of outbreak and emergency response.
A key component of these efforts is related to the IHR. To build resilience, the
implementation of the IHR should be approached as a holistic process and should also be
embedded in national policy development and planning, legislative action, and regulatory
frameworks. The implementation should include improvements in health services
organization to support infection prevention and control, strengthening of health
surveillance networks and laboratory capacity, and development of health workforce
competencies in the areas of outbreak and emergency response.
The policy also calls for ensuring reserve capacity, with the necessary supply of appropriate health workers, financing, medicines, and health technologies to allow a rapid scale-up of health services during acute or sustained health events.
The resilient health systems policy was reviewed by the 158th Session of the Executive Committee of PAHO in June 2016 and incorporated into the agenda for the 55th Directing Council.
 The Bureau continued to provide technical cooperation to assist countries in
advancing their implementation of the regional Strategy for Universal Access to Health
and Universal Health Coverage (CD53/5), approved by PAHO’s 53rd Directing Council
in 2014. Areas of focus for the Bureau’s work during 2015–2016 included health system
financing, health-related legislation, human resources for health, and regulatory systems.
The Bureau continued to provide technical cooperation to assist countries in
advancing their implementation of the regional Strategy for Universal Access to Health
and Universal Health Coverage (CD53/5), approved by PAHO’s 53rd Directing Council
in 2014. Areas of focus for the Bureau’s work during 2015–2016 included health system
financing, health-related legislation, human resources for health, and regulatory systems.
The regional strategy recommends a benchmark of 6% of GDP as the minimum
level of health spending needed for countries to achieve universal health. To help
countries reach this recommended goal, the Bureau led a participatory process to develop
a conceptual framework for efforts to improve efficiency in health systems financing and
generate additional resources for health.
As part of this process, the Bureau commissioned a regional study on fiscal space
for health, which analyzed the experiences of 14 countries in the Region. The results of
the study were incorporated into a working document titled Fiscal space for increasing health priority in public spending in the Americas Region. This document presents
evidence that countries can successfully generate new resources for health to achieve the
6% GDP threshold for universal health. Based on the experiences analyzed, the report
recommends that additional resources be mobilized from domestic sources in order to
ensure sustainability. Effective methods of generating these resources include new or
increased taxes, especially on products that are harmful to health; improved efficiency in
health systems and public health spending; and improved tax administration. The report
recommends that countries seeking to create new fiscal space promote an all-inclusive
dialogue on health as a social right and on the need for additional resources to develop
systems for universal health.
The conceptual framework was presented at a regional forum titled “Universal
Health: An Indispensable Investment for Sustainable Human Development,” which was
convened at PAHO Headquarters in December 2015. Meeting participants included
representatives from ministries of health and social security systems of Member States, as
well as the World Bank, the International Monetary Fund (IMF), the OAS, the UN
Economic Commission for Latin America and the Caribbean (ECLAC), and academic
institutions in the Region.
In related efforts, Bureau staff worked with WHO counterparts to implement a
survey in Peru on budgeting practices for health. The survey is part of an initiative of the
Organization for Economic Cooperation and Development (OECD) Health Committee
Joint Network on the Fiscal Sustainability of Health Systems. The initiative seeks to
ensure that the provision of public resources for health systems is aligned with sectoral
efforts to promote equity and efficiency and to sustain progress toward universal health.
The survey aims to provide an internationally comparable set of data that will allow for
analysis and benchmarking of good practices in budgeting for health.
 In October 2015, PAHO’s 54th Directing Council approved a new regional
Strategy on Health-related Law (Document CD54/14, Rev. 1). That strategy promotes the
development, implementation, and revision of laws and legal frameworks to protect
human rights, advance health equity, promote solidarity and nondiscrimination in health,
reduce risk factors, and address the social determinants of health. Developed through a
consultative process led by the Bureau and a working group of Member States, the
strategy calls for the exchange of strategic information, such as best practices and judicial
decisions, among Members States and with international organizations, and for stronger
coordination and collaboration at the national level between health authorities and the
legislative branch.
In October 2015, PAHO’s 54th Directing Council approved a new regional
Strategy on Health-related Law (Document CD54/14, Rev. 1). That strategy promotes the
development, implementation, and revision of laws and legal frameworks to protect
human rights, advance health equity, promote solidarity and nondiscrimination in health,
reduce risk factors, and address the social determinants of health. Developed through a
consultative process led by the Bureau and a working group of Member States, the
strategy calls for the exchange of strategic information, such as best practices and judicial
decisions, among Members States and with international organizations, and for stronger
coordination and collaboration at the national level between health authorities and the
legislative branch.
During the review period, the Bureau organized a series of country consultations for the purpose of identifying priorities for strengthening human resources to support progress toward universal health. The consultations produced three main strategic orientations: (1) strengthening the leadership and governance capacity of national authorities responsible for human resources for health; (2) guaranteeing equity in staffing and distribution of human resources for health; and (3) reorienting the training of certain categories of health professionals who are being proposed for placement in primary health care settings. These perspectives were discussed and endorsed by Member State representatives at a regional meeting on human resources for health that was held in Buenos Aires during September 2015 and that focused on new and renewed challenges for health teams.
As part of its technical cooperation to strengthen regulatory capacity in PAHO
Member States, the Bureau continued to support the Caribbean Regulatory System (CRS), a subregional initiative to create a single entry point into the pharmaceutical
market of the Caribbean Community (CARICOM). Based on CARICOM’s longstanding
tradition of addressing health issues through collaborative action, the CRS provides an
innovative approach intended to harness the limited resources within that subregion,
foster convergence and transparency, and create a market incentive for manufacturers to
offer products to all CARICOM states.
 Launched in late 2015 as a pilot project based within the Caribbean Public Health
Agency (CARPHA), the CRS has focused initially on registering priority essential
generic medicines through an abbreviated review procedure and on leveraging
information on product registration and postmarketing surveillance provided by national
regulatory authorities of regional reference (NRAr) in PAHO Member States, but while
still retaining sovereignty over decision-making.
Launched in late 2015 as a pilot project based within the Caribbean Public Health
Agency (CARPHA), the CRS has focused initially on registering priority essential
generic medicines through an abbreviated review procedure and on leveraging
information on product registration and postmarketing surveillance provided by national
regulatory authorities of regional reference (NRAr) in PAHO Member States, but while
still retaining sovereignty over decision-making.
To ensure that the registration of products is followed by a rapid uptake by
CARICOM Member States, the Bureau partnered with Health Canada and the Caribbean
Law Institute Centre in Barbados to develop a template for memoranda of understanding
between the CRS and participating Member States. The Bureau also provided capacitybuilding
opportunities, such as a workshop on good review practices for CARICOM, held
in Trinidad and Tobago in March 2016. The national regulatory authorities of Argentina,
Brazil, Canada, Colombia, Cuba, Mexico, and the United States also provided substantial
support for these efforts.
The Bureau facilitated the initial conception and design of the CRS and has
supported its implementation since it was formally approved by CARICOM ministers of
health at the 2014 meeting of the CARICOM Council on Human and Social
Development (COHSOD). In addition to providing technical cooperation, the Bureau
successfully mobilized resources from partners, including the U.S. Food and Drug
Administration (U.S. FDA), Health Canada, and the Bill and Melinda Gates Foundation.
As the CRS’s implementation progresses, the proposed organizational model will seek
financial sustainability through a system of user fees.
In other efforts to strengthen national regulatory capacity, the Bureau facilitated
the establishment of a new regional mechanism for harmonization and convergence on
the exchange of medical devices reports, through which regulatory authorities monitor
the performance and safety of medical devices postmarket. As part of the initiative, the
national regulatory authorities of 14 Member States agreed on a plan for additional
collaborative action in this area during 2016.
 As part of its efforts to support progress in advancing the Sustainable
Development Goals, health equity, and particularly universal health, the Bureau launched
a major new initiative in 2016 to provide guidance and generate evidence for PAHO
Member States to address the social determinants of health and the cross-cutting themes
of gender, ethnicity, and human rights. This new Commission on Equity and Health
Inequalities in the Region of the Americas was launched at a high-level event at PAHO
Headquarters in May 2016.
As part of its efforts to support progress in advancing the Sustainable
Development Goals, health equity, and particularly universal health, the Bureau launched
a major new initiative in 2016 to provide guidance and generate evidence for PAHO
Member States to address the social determinants of health and the cross-cutting themes
of gender, ethnicity, and human rights. This new Commission on Equity and Health
Inequalities in the Region of the Americas was launched at a high-level event at PAHO
Headquarters in May 2016.
The commission brings together more than a dozen leading international experts
on health policy and social determinants of health with a mandate to undertake the
Review of Equity and Health Inequalities in the Americas, the first such comprehensive
effort to gather evidence on health inequities in the Region. The commission will
investigate how socioeconomic factors, structural factors, and identity influence health in
the countries of the Americas. The group will make concrete recommendations for action
to reduce or eliminate persisting health equity gaps in the Region. The commission’s
interim report is expected to be published in early 2017, with a final report in 2018.
In addition to its support for the Zika response and other health emergencies, the
Bureau continued to provide technical cooperation and coordination for country efforts to
advance the elimination of other infectious diseases. During the period under review,
notable progress was recorded in regional efforts to eliminate measles and mother-to-child
transmission of HIV and syphilis, and in support of global efforts to eradicate
poliomyelitis.
In the case of measles, the Region’s most recent outbreak, in Ceará, Brazil, ended
in July 2015, less than three months after the International Expert Committee for
Documenting and Verifying Measles, Rubella, and Congenital Rubella Syndrome in the
Americas (IEC) had met and declared the Americas free of transmission of rubella and
congenital rubella syndrome (CRS). Although the ongoing outbreak in Brazil had
prevented the IEC from making a similar declaration regarding measles, by December
2015 they were able to review the evidence and confirm that the 20-month-long measles
outbreak in Ceará had been interrupted. This evidence included those established
laboratory, surveillance, and vaccination coverage data that had been defined as essential
criteria for elimination. However, the verification process required the IEC to wait
another full 12 months after 6 July 2015, the onset date of the last recorded endemic
measles case, to be sure that no additional cases were confirmed in Brazil during that
period.
To support the IEC’s work, the Bureau requested that PAHO Member States
provide updates of their measles elimination reports for 2012–2015. As of June 2016,
Bureau staff were compiling the country submissions into a regional report for
presentation at an August 2016 meeting of IEC members with technical staff from the
ministries of health and the chairpersons of countries’ national verification commissions.
The meeting would review the updated evidence and determine the feasibility of
declaring the Americas free of measles in September 2016.
 Having been, in 1991, the first WHO region to eliminate poliomyelitis, the
Region of the Americas has continued to support global polio eradication efforts.
Currently this is being done under the Polio Eradication and Endgame Strategic Plan
2013–2018 of the Global Polio Eradication Initiative, which was endorsed by the World
Health Assembly.
Having been, in 1991, the first WHO region to eliminate poliomyelitis, the
Region of the Americas has continued to support global polio eradication efforts.
Currently this is being done under the Polio Eradication and Endgame Strategic Plan
2013–2018 of the Global Polio Eradication Initiative, which was endorsed by the World
Health Assembly.
A central component of the Endgame Plan is the eventual global withdrawal of all
oral polio vaccine (OPV). This is to be done through a phased process in which countries
that are currently using OPV switch from trivalent OPV (covering poliovirus types 1, 2,
and 3) to bivalent OPV (types 1 and 3), and all countries that are not yet using inactivated
polio vaccine (IPV) introduce at least one dose of IPV into their routine immunization
schedules. Between February 2015 and April 2016, the 32 countries and territories in the
Region that were not already using IPV introduced it into their schedules, and 36 made
the switch from trivalent to bivalent OPV as part of the global switchover in April 2016.
A second key component of the Endgame Plan is the destruction or containment
of all polioviruses type 2 (wild, Sabin, and vaccine-derived poliovirus) at essential
facilities. The regional plan also called for the containment of wild poliovirus types 1 and
3. By June 2016, 33 PAHO Member States, six U.K. territories, and three Associate
Members had submitted reports on the destruction or containment of all wild and
vaccine-derived poliovirus. All PAHO Member States were expected to submit reports on
their containment of Sabin type 2 polioviruses by August 2016.
 In mid-2015, immediately following Cuba’s success in becoming the first country
in the world to be validated as having eliminated mother-to-child transmission (MTCT)
of HIV and syphilis, several other countries and territories of the Americas officially
requested that the Bureau initiate the process for validating their achievement of this dual
elimination. By the end of 2015, 17 countries and territories in the Region were reporting
data consistent with the dual elimination targets. With a new cluster validation approach
under way in the Caribbean, at least 10 countries and territories were expected to be
validated before the end of 2016.
In mid-2015, immediately following Cuba’s success in becoming the first country
in the world to be validated as having eliminated mother-to-child transmission (MTCT)
of HIV and syphilis, several other countries and territories of the Americas officially
requested that the Bureau initiate the process for validating their achievement of this dual
elimination. By the end of 2015, 17 countries and territories in the Region were reporting
data consistent with the dual elimination targets. With a new cluster validation approach
under way in the Caribbean, at least 10 countries and territories were expected to be
validated before the end of 2016.
The elimination efforts that led to these achievements were carried out within the
framework of the 2010 PAHO Strategy and Plan of Action for the Elimination of Motherto-
Child Transmission of HIV and Congenital Syphilis. These efforts have had a direct
impact on perinatal HIV and congenital syphilis. In addition, they have strengthened
maternal and child health programs, health information systems, and accountability by
Member States, with an increased number of countries providing yearly data to enable the
Bureau to monitor their achievements.
Given these positive impacts and recognizing synergies between these efforts and
related public health programs and initiatives, the Bureau began developing a second
phase of the dual elimination initiative to target other mother-to-child transmitted
infections, such as hepatitis B and Chagas disease. The expanded initiative will promote
intensified MTCT elimination efforts as an integral part of the Sustainable Development
Goals (SDGs).
The Bureau continued its support of Member States’ efforts to combat
noncommunicable diseases (NCDs), which are the leading cause of morbidity, mortality,
and disability throughout the Americas. According to data from the PAHO Mortality
Database, NCDs are responsible for 80% of all deaths in the Region, and 35% of these
deaths are premature, that is, occurring in persons aged 30 to 69 years.
During the year under review, Bureau staff undertook a midterm review of the
implementation of the regional Plan of Action for Prevention and Control of
Noncommunicable Diseases (Document CD52/7, Rev. 1), which aims to reduce NCD
deaths in the Americas by 15% by 2019. The assessment report noted a stable pattern or a
modest decline in premature NCD mortality in nearly all of the countries and territories
of the Region. However, fewer than half appeared to be on track to meet the 15%
reduction target, with the slowest progress being observed in Central America and the
Caribbean.
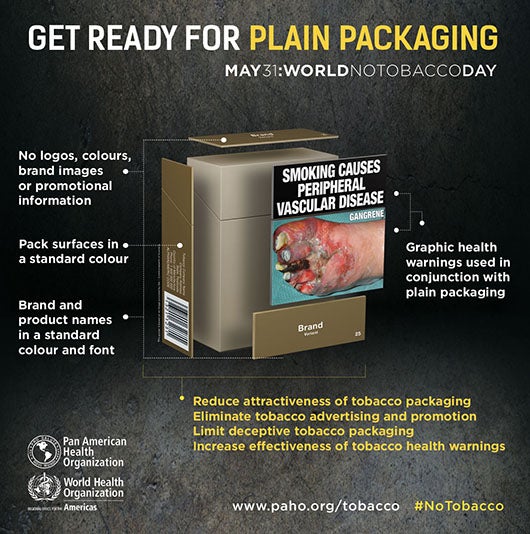 On the basis of survey responses from ministries of health in 38 countries and
territories in the Region, the review found that intensified action was needed in key areas,
particularly with the development of national NCD plans and creation of multisectoral
commissions. The report also urged stepped-up implementation of the WHO Framework
Convention on Tobacco Control (FCTC) and other interventions to reduce consumption
of harmful products, such as new or increased taxes on alcohol and sugary beverages,
restrictions on the marketing of ultraprocessed foods and beverages to children, and
limitations on the marketing of breast milk substitutes.
On the basis of survey responses from ministries of health in 38 countries and
territories in the Region, the review found that intensified action was needed in key areas,
particularly with the development of national NCD plans and creation of multisectoral
commissions. The report also urged stepped-up implementation of the WHO Framework
Convention on Tobacco Control (FCTC) and other interventions to reduce consumption
of harmful products, such as new or increased taxes on alcohol and sugary beverages,
restrictions on the marketing of ultraprocessed foods and beverages to children, and
limitations on the marketing of breast milk substitutes.
In addition, the assessment report urged countries to utilize the PAHO Strategic
Fund to access essential medicines for NCDs, especially those for blood pressure control
and the prevention of cardiovascular disease. The report was reviewed in June 2016 by
the 158th Session of the Executive Committee of PAHO and recommended for inclusion
on the agenda of the 55th Directing Council.
To generate evidence for policy-making and resource allocation for NCDs in the
Americas, the Bureau partnered with Harvard University’s T.H. Chan School of Public
Health to adapt and apply WHO’s EPIC model for estimating the impact of diseases on
aggregate economic output. The collaboration applied Harvard’s EPIC-H Plus model,
which takes account of morbidity (specifically, the disability-adjusted life years
(DALYs)) and the costs of health spending for NCDs and mental conditions.
The model was initially applied in Costa Rica, Jamaica, and Peru. It estimated that
if no action was taken, lost GDP between 2015 and 2030 as a result of NCDs would be equivalent to 142% of Costa Rica’s 2013 GDP, 106% of Jamaica’s 2013 GDP, and 243%
of Peru’s 2013 GDP. These and other findings of the collaboration were presented at the
World Economic Forum on Latin America in Medellin, Colombia, in June 2016. The
collaboration also includes the development of guidelines for countries in the Americas to
calculate similar estimates on their own, using the EPIC-H Plus model, which will be
made available free of charge as a public good.
One of the key action areas defined in the regional NCD plan is the use of fiscal
policy to reduce NCD risk factors by “making the healthy choice the easy choice.”
Increasing excise taxes on harmful products can discourage their consumption and thus
decrease health costs, while also generating new revenues that can be used for health and
development. To build country capacity to apply such an approach, the Bureau organized
subregional workshops in Central and South America, where officials from ministries of
health and finance worked together to develop fiscal scenarios for amending their tobacco
tax structures to meet both health and revenue targets. The workshops led to the
development of proposals for tobacco tax increases in Colombia and Peru and to the
creation of subregional networks to discuss tax harmonization and strategies to eliminate
illicit trade in tobacco products.
 In taking advantage of fiscal policy for improving the prevention and control of
NCDs and their risk factors, the Bureau in 2015 published a report that documents
Mexico’s experience with the adoption of a 10% excise tax on sugar-sweetened beverages
in 2014. This report, titled Taxes on Sugar-sweetened Beverages as a Public Health
Strategy: The Experience of Mexico, describes how supporters of the tax initiative were
able to overcome the active opposition of the soft drink industry and its allies. The report
cites preliminary results of a study conducted jointly by Mexico’s National Institute of
Public Health (INSP) and the University of North Carolina at Chapel Hill (USA), which
revealed an average 6% reduction in sales of the taxed beverages during the tax’s first
year, as compared with the previous year. Mexico’s experience has informed similar
efforts in Barbados, Dominica, and Ecuador, which all implemented soda taxes in 2015
or 2016.
In taking advantage of fiscal policy for improving the prevention and control of
NCDs and their risk factors, the Bureau in 2015 published a report that documents
Mexico’s experience with the adoption of a 10% excise tax on sugar-sweetened beverages
in 2014. This report, titled Taxes on Sugar-sweetened Beverages as a Public Health
Strategy: The Experience of Mexico, describes how supporters of the tax initiative were
able to overcome the active opposition of the soft drink industry and its allies. The report
cites preliminary results of a study conducted jointly by Mexico’s National Institute of
Public Health (INSP) and the University of North Carolina at Chapel Hill (USA), which
revealed an average 6% reduction in sales of the taxed beverages during the tax’s first
year, as compared with the previous year. Mexico’s experience has informed similar
efforts in Barbados, Dominica, and Ecuador, which all implemented soda taxes in 2015
or 2016.
As part of its support for the NCD regional action plan, the Bureau has also been
assisting countries to use population-based surveys to strengthen their surveillance of
NCDs, their risk factors, and mental health disorders. In 2016 the Bureau piloted the use
of two new technologies for collecting data through household surveys: Web-based tablet
computers for data entry and devices for biochemical measurements that allow more
accurate monitoring of cholesterol levels and diabetes. These were used in Ecuador to
implement the WHO STEPS survey at a subnational level. These new technologies
received positive reviews from both the Ministry of Health and the National Statistics
Office. The Bureau has procured a stock of these devices that it will rotate among other
Member States for use in these national surveys.
In support of NCD surveillance, the Bureau further developed a technical package
to assess country capacity to monitor NCDs and their risk factors, identify gaps in
monitoring, establish priorities, and track progress toward global and regional NCD
commitments. The package was applied in countries of the Andean Subregion in October
2015 and provided useful input for the development of plans to improve countries’ NCD
surveillance capacity. The Bureau plans to use this package in other subregions.
 During the review period, the Bureau provided technical expertise and enhanced
support to its Member States in the area of gender, ethnicity, and health. Its work in the
specific area of gender and health was enabled by a new Strategy and Plan of Action on
Strengthening the Health System to Address Violence against Women, which was
approved by PAHO’s 54th Directing Council in October 2015. This framework, which
was the first of its kind in any WHO region, takes a public health approach to the
problem of violence against women and provides a roadmap for health systems to play a
key role in multisectoral efforts to prevent and respond to such violence. It was
developed through a participatory process involving more than 100 representatives from
governments, civil society, academic institutions, multilateral organizations, and UN
agencies.
During the review period, the Bureau provided technical expertise and enhanced
support to its Member States in the area of gender, ethnicity, and health. Its work in the
specific area of gender and health was enabled by a new Strategy and Plan of Action on
Strengthening the Health System to Address Violence against Women, which was
approved by PAHO’s 54th Directing Council in October 2015. This framework, which
was the first of its kind in any WHO region, takes a public health approach to the
problem of violence against women and provides a roadmap for health systems to play a
key role in multisectoral efforts to prevent and respond to such violence. It was
developed through a participatory process involving more than 100 representatives from
governments, civil society, academic institutions, multilateral organizations, and UN
agencies.
The 54th Directing Council also endorsed new priorities for advancing gender
equality in health, based on the Report of the Evaluation of the Gender Equality Plan of
Action 2009-2014. The report analyzes progress as well as the remaining challenges to
advancing gender equality based on country self-assessments, and it presents new lines of
action to guide the Bureau’s future work in this area. They include conducting research
and applying innovative methodologies to address gender inequities in health, generating
sector-specific evidence and gender analysis, and addressing emerging themes such as the
health concerns of the lesbian, gay, bisexual, and trans (LGBT) communities as well as
masculinities.
To promote the generation of evidence, the Bureau published the sixth edition of
the statistical compendium titled “Gender, Health and Development in the Americas.” In
April 2016 the Bureau convened an expert group to review a core set of indicators on
gender and health for the Americas as a whole, within the frameworks of universal health
and the SDGs. Bureau experts also provided inputs on a set of gender equality indicators
for the Caribbean (coordinated by UN Women), which were presented to Dominica,
Guyana, Jamaica, and Suriname for consultation and later to CARICOM for validation
and eventual adoption by the Caribbean countries.
Under PAHO’s universal health mandate, Bureau staff worked with the Women’s
Health Network for Latin America and the Caribbean (RSMLAC for its acronym in
Spanish) to promote a debate on universal health and its implications for women. The
Bureau also provided guidance and inputs on gender and universal health for the Technical Commission on Gender and Health in Central America, a subgroup of the
Health Commission of the Council of Ministers of Health of Central America
(COMISCA). In Jamaica, the Bureau supported training for Ministry of Health staff on
these issues as part of the country’s efforts to achieve universal health.
 The Bureau also provided support at the national, subregional, and regional levels
to assist countries in addressing the noticeable ethnic inequities in health that persist
throughout the Region. For example, in the South American Chaco region, technical
support was provided for a workshop entitled “United to Action, Moving towards
Universal Health in South American Chaco.” Representatives from the ministries of
health and the indigenous peoples participated in this forum, an important outcome of
which was the formulation of an inter-country project to advance universal health
coverage among indigenous peoples. Moreover, it is important to highlight that at the
global level, PAHO has also played a key interagency role as co-chair of the UNDGLAC
Interagency Group on Indigenous Peoples. A priority focus of this group as well as
the Bureau’s technical cooperation is improving the availability and quality of data on
ethnicity and health.
The Bureau also provided support at the national, subregional, and regional levels
to assist countries in addressing the noticeable ethnic inequities in health that persist
throughout the Region. For example, in the South American Chaco region, technical
support was provided for a workshop entitled “United to Action, Moving towards
Universal Health in South American Chaco.” Representatives from the ministries of
health and the indigenous peoples participated in this forum, an important outcome of
which was the formulation of an inter-country project to advance universal health
coverage among indigenous peoples. Moreover, it is important to highlight that at the
global level, PAHO has also played a key interagency role as co-chair of the UNDGLAC
Interagency Group on Indigenous Peoples. A priority focus of this group as well as
the Bureau’s technical cooperation is improving the availability and quality of data on
ethnicity and health.
In an effort to advance the health of Afro-descendant populations, the Bureau
participated in a regional technical meeting on Afro-descendant health that was convened
in Cartagena, Colombia, in November 2015. This meeting provided a unique opportunity
to identify and address Afro-descendants’ health priorities and challenges and to design
and implement effective health policies and programs with an intercultural perspective.
As a follow-up to this meeting, and within the context of the International Decade for
People of African Descent, the Bureau has been actively providing technical support for
the development of subregional plans of action for the health of Afro-descendants.
In December 2015, a Regional Meeting on Ethnicity and Health was convened for
ministers of health together with regional and international experts as well as
representatives of indigenous and Afro-descendant populations for the purpose of
highlighting and discussing key issues to address ethnic disparities in health. This
meeting served as a platform to discuss the next steps towards developing a Strategy and
Action Plan on Ethnicity and Health, as well as to facilitate the dialogue of the United
States-Brazil Joint Action Plan to Eliminate Racial and Ethnic Discrimination. Moreover,
under the umbrella of the International Decade for People of African Descent, the Bureau
provided a report containing the most relevant health findings in the Region to the UN
Working Group of Experts on People of African Descent. In May 2016, the Bureau
organized two side events during the 15th session of the UN Permanent Forum on
Indigenous Issues in New York, in order to highlight current challenges to advancing the
right to health of indigenous peoples.
The Bureau’s work in the area of gender, ethnicity, and health was notably
strengthened through enhanced interprogrammatic collaboration across and between
those technical units responsible for tuberculosis, malaria, disaster risk reduction, and mental health. For example, a technical meeting was conducted with the Baramita
community in Guyana to address mental health issues among the indigenous residents,
and a plan of action was developed based on these emerging discussions. In Ecuador,
technical cooperation was provided to reduce maternal mortality with an emphasis on the
country’s indigenous peoples.
 Several initiatives and developments at the institutional level contributed to
enhancing the efficiency, effectiveness, and accountability of the Bureau’s operations
during the year under review. One of the most noteworthy was the joint end-of-biennium
assessment by the Bureau and PAHO Member States, the first process of this kind to be
undertaken in any WHO region. This exercise provided a unique opportunity for the
Bureau and ministries of health of Member States to jointly review public health gains,
gaps, challenges, opportunities, and lessons learned from the delivery of the biennial
work plan 2014-2015. This assessment covered the implementation of the 2014-2015
Program and Budget and the first two years of the current Strategic Plan. The assessment
was facilitated by the rollout of the Bureau’s new strategic plan monitoring system,
developed in collaboration with and formally approved by Member States as part of the
PAHO Strategic Plan 2014-2019.
Several initiatives and developments at the institutional level contributed to
enhancing the efficiency, effectiveness, and accountability of the Bureau’s operations
during the year under review. One of the most noteworthy was the joint end-of-biennium
assessment by the Bureau and PAHO Member States, the first process of this kind to be
undertaken in any WHO region. This exercise provided a unique opportunity for the
Bureau and ministries of health of Member States to jointly review public health gains,
gaps, challenges, opportunities, and lessons learned from the delivery of the biennial
work plan 2014-2015. This assessment covered the implementation of the 2014-2015
Program and Budget and the first two years of the current Strategic Plan. The assessment
was facilitated by the rollout of the Bureau’s new strategic plan monitoring system,
developed in collaboration with and formally approved by Member States as part of the
PAHO Strategic Plan 2014-2019.
The joint assessment began in November 2015 with country-level assessments,
which were subsequently validated by Bureau technical staff and consolidated into
Organization-wide results. The findings of the assessment showed that 90% of the
outcome indicators of the PAHO Strategic Plan were on track to be achieved by 2019,
and all 114 expected outputs in the 2014-2015 Program and Budget were either fully or
partially achieved. The full findings were presented in the Preliminary Report of the Endof-
Biennium Assessment of the Program and Budget 2014-2015/First Interim Report on
the PAHO Strategic Plan 2014-2019, which was reviewed by the 158th Session of the
Executive Committee in June 2016.
There was 100% participation in the conduct of this first joint assessment, with all
51 countries and territories of the Region involved. PAHO Member States have
recommended that the experience be shared with WHO and other WHO regions as a best
practice in transparency, accountability, and reporting.
The ongoing process of WHO reform, launched in 2011 in response to the
changing needs for global public health leadership, continued to advance during the
reporting period. Several critical issues for PAHO and its Member States related to WHO
reforms were included on the agenda of the 69th World Health Assembly. In anticipation
and in preparation for this development, PAHO’s Director initiated and maintained a
close dialogue with Member States during the months preceding the assembly in order to
keep them fully apprised of new developments; to seek their feedback on critical issues;
and to provide advice on the process and its implications for PAHO as the Regional
Office for the Americas of WHO. This proactive engagement included briefings during
the PAHO Sub-regional Managers’ Meetings, PAHO Governing Bodies meetings, and
additional virtual sessions, together with support for several PAHO Member States that
played pivotal roles in the negotiations at the global level by leading or participating in
WHO Working Groups.
Many of the reforms contemplated by WHO had already been adopted by PAHO
in recent years, even prior to their implementation at WHO’s Headquarters. For example,
PAHO had already introduced reforms in the programmatic, managerial, and budget
areas. During this review period, three issues of particular importance to PAHO and its
Member States were advanced: a) proposed changes in WHO’s governance structure,
b) the proposed new Framework on Engagement with non-State Actors (FENSA), and
c) the creation of a new WHO Health Emergencies Program.
These three proposals included new lines of command and accountability and
raised the real possibility of serious challenges to PAHO’s constitutional framework.
That framework, which establishes PAHO as the specialized agency for health within the
inter-American system, also affirms PAHO as an independent entity, which along with its
Director, is directly accountable to the Member States of the Americas. Considering this
special status, which is unique among WHO regional offices, the Bureau and PAHO
Member States provided input into the negotiations that emphasized the importance of
aligning PAHO with the new reforms in ways that would strengthen decision-making and
transparency at the global level, while simultaneously respecting PAHO’s status as an
independent international organization.
 With regard to the WHO governance reforms, PAHO Member States supported
proposed recommendations for increased regional coordination with WHO in the areas of
management, transparency, and alignment, but also underscored the importance of
regional specificities.
With regard to the WHO governance reforms, PAHO Member States supported
proposed recommendations for increased regional coordination with WHO in the areas of
management, transparency, and alignment, but also underscored the importance of
regional specificities.
In negotiations on FENSA, delegates from the Americas emphasized that PAHO
has since 2005 had its own guidelines for collaborating with non-State actors, including
the private sector, NGOs, foundations, and academic institutions. On the understanding
that FENSA would provide an additional means to ensure engagement with non-State
actors utilizing a principled, implementable, and balanced approach without
compromising the Organization’s independence, credibility, or reputation, the Bureau
will submit FENSA to PAHO’s Governing Bodies for consideration in accordance with
PAHO’s legal status.
Regarding the new WHO Health Emergencies Program, PAHO Member States
presented a joint statement to the 69th World Health Assembly expressing their support
for the new program, while acknowledging that PAHO’s Department of Emergency
Preparedness and Disaster Relief has been extremely effective in responding to
emergencies and disasters in the Region of the Americas, based on experiences gained,
the expertise honed, and the lessons learned since 1976. The countries emphasized that
their support for the new WHO Health Emergencies Program assumes that PAHO’s
regional program will continue to respond fully to the needs of Member States in the
Americas, while working in a coordinated manner with the WHO program.
Proactive input by the Bureau and PAHO Member States had a favorable impact
on the outcomes of the 69th World Health Assembly, which included approval of a
consensually agreed FENSA, the creation of the new WHO Health Emergencies
Program, and WHO governance reforms. PAHO’s Governing Bodies are expected to
further discuss these actions and the regional ramifications at the meeting of the
55th PAHO Directing Council, 68th Session of the Regional Committee of WHO for the
Americas, in September 2016.
 Both the Revolving Fund for Vaccine Procurement (created in 1977) and the
Strategic Fund for Public Health Supplies (created in 2002) have been highly successful
since their inception in using pooled procurement at the regional level to improve access
for PAHO Member States to vaccines, other medicines, and health supplies.
Both the Revolving Fund for Vaccine Procurement (created in 1977) and the
Strategic Fund for Public Health Supplies (created in 2002) have been highly successful
since their inception in using pooled procurement at the regional level to improve access
for PAHO Member States to vaccines, other medicines, and health supplies.
A new agreement signed in March 2015 with the Global Fund to Fight AIDS,
Tuberculosis and Malaria positioned the Bureau to substantially scale up its procurement
of high-cost medicines on behalf of PAHO Member States. This new agreement gives the
PAHO Strategic Fund access to the Global Fund’s Pooled Procurement Mechanism.
Similar to PAHO’s Strategic Fund and Revolving Fund, the Global Fund mechanism uses
pooled procurement to access medicines and medical supplies at lower prices by
consolidating demand, but in this case, on a global scale.
The Global Fund’s Pooled Procurement Mechanism addresses key procurementrelated
issues, including ensuring the uninterrupted availability of supplies, product
quality and safety, and negotiation of attractive prices. Under this new agreement, the
Strategic Fund has obtained medicines at appreciably reduced prices, in some cases as
low as one-tenth the price paid previously by PAHO Member States. This new
arrangement is also expected to result inter alia in improved vendor performance, as this
will be jointly measured by both the Bureau and the Global Fund, as well as more rapid
access to attractively priced medicines during situations of urgent need.
 The Bureau continued to advance the implementation of the new PASB
Management Information System (PMIS) during the course of the year under review.
Phase 2 of the system, covering financial, budget, and procurement functions, was tested
between July and November 2015, with staff training taking place from September
through December. As scheduled, Phase 2 was fully activated in early January 2016,
complete with a Post-Go-Live Center that provided support to all staff throughout the
Bureau. Rollout of the second phase of PMIS has presented challenges, and it has
required a substantial investment of time and effort on the part of many Bureau staff.
Overall, however, the rollout has been accomplished with relatively minimal disruption
to the implementation of the Bureau’s technical programs and day-to-day operations,
even though some issues are yet to be resolved and training is ongoing. This experience is in marked contrast to that of many large international organizations when implementing
new enterprise resource planning systems and is a credit to the good planning and
extraordinary efforts of the Bureau staff involved.
The Bureau continued to advance the implementation of the new PASB
Management Information System (PMIS) during the course of the year under review.
Phase 2 of the system, covering financial, budget, and procurement functions, was tested
between July and November 2015, with staff training taking place from September
through December. As scheduled, Phase 2 was fully activated in early January 2016,
complete with a Post-Go-Live Center that provided support to all staff throughout the
Bureau. Rollout of the second phase of PMIS has presented challenges, and it has
required a substantial investment of time and effort on the part of many Bureau staff.
Overall, however, the rollout has been accomplished with relatively minimal disruption
to the implementation of the Bureau’s technical programs and day-to-day operations,
even though some issues are yet to be resolved and training is ongoing. This experience is in marked contrast to that of many large international organizations when implementing
new enterprise resource planning systems and is a credit to the good planning and
extraordinary efforts of the Bureau staff involved.
In parallel with the implementation of PMIS, the Bureau also embarked upon the
rollout of the new Information Technology Strategy that was approved in 2015. The IT
strategy defines five focus areas: technical program support, country office support,
PMIS, IT innovation, and maintaining stable IT operations. Highlights of the strategy’s
implementation included the deployment of a new invoice verification system for the
Mais Médicos Project in Brazil; improvements in the PAHO Mortality Database; an
assessment of the PAHO Health Information Platform (PLISA for its acronym in
Spanish); and the implementation of a vaccine surveillance tool and a new dengue
application that will allow countries to transmit their surveillance data to PLISA using a
standardized format.
In addition, in country offices, Internet bandwidth was increased in order to
enhance the offices’ connectivity both to the cloud and with Headquarters’ systems,
telephone and server infrastructures were upgraded, and a number of desktop computers
were replaced. In the area of innovation, a new cloud-based Service Request System was
instituted to support all users and systems across the Bureau, and a new tablet-based
paperless system was created for conferences and meetings. This new modality was
introduced to PAHO’s Governing Bodies at the 158th session of the Executive
Committee.
Additionally, Internet security at headquarters was improved, with new software
for Web content monitoring and filtering and with last-generation firewalls. Finally,
network upgrades were made to improve data speed and application response, with
additional improvements in backup systems and data recovery.
 An important development in the area of human resources was the launch in July
2015 of the Respectful Workplace Initiative, a Bureau effort to foster a climate of respect
and inclusion throughout the institution. Led by the Office of the Ombudsman, the
initiative builds on earlier efforts spearheaded by the PAHO Staff Association in 2012,
the department of Human Resources Management (HRM) in 2013, and the “Make
Kindness Contagious” campaign led by the Special Program on Sustainable Development
and Health Equity (SDE) in 2014.
An important development in the area of human resources was the launch in July
2015 of the Respectful Workplace Initiative, a Bureau effort to foster a climate of respect
and inclusion throughout the institution. Led by the Office of the Ombudsman, the
initiative builds on earlier efforts spearheaded by the PAHO Staff Association in 2012,
the department of Human Resources Management (HRM) in 2013, and the “Make
Kindness Contagious” campaign led by the Special Program on Sustainable Development
and Health Equity (SDE) in 2014.
Developed through a participatory process that included interdepartmental
working groups with representation from the PAHO Staff Association, the initiative set
out three overarching objectives: a) raise awareness and promote the importance of
individual responsibility in creating a respectful working environment; b) provide
information and basic tools needed to create a culture of respect that encourages trust,
responsibility, accountability, mutual respect, and open communication, while embracing the dignity and diversity of individuals; and c) increase knowledge among staff about the
different mechanisms and instances available to address workplace concerns.
Implementation of the initiative’s first phase began in 2015-2016. It included the
development of an awareness campaign designed to encourage staff to contribute to an
overall productive and supportive working environment, with an emphasis on prevention
and early resolution of conflict using available institutional resources. In collaboration
with the Bureau’s Ethics Office, a new e-learning course was developed that focuses on
the principles defined in the Bureau’s Code of Ethical Principles and Conduct, Policy on
the Prevention and Resolution of Harassment in the Workplace, and Zero Tolerance for
Fraud and Corruption policy, among others. The aim of the course is to illustrate how
mutual respect and collegial working relations can produce a more effective and
productive work environment.
In a related effort, the Ethics Office developed a new survey on the Bureau’s
ethical climate and work environment. The survey is designed to assess the work
environment in different Bureau offices and to gauge awareness among staff about the
policies and resources that constitute the PAHO Integrity and Conflict Management
System (ICMS). This survey also measures how comfortable people feel in working at
PAHO and whether they feel that their work is valued. The results of the survey will be
used to identify specific areas of concern that require additional attention.
 The period under review, from mid-2015 through mid-2016, was filled both with
numerous achievements of which the Bureau can be justifiably proud as well as with
numerous challenges, some of which have been successfully resolved while others are
works in progress. Under the rubric of achievements, we responded vigorously and
provided strong leadership and dynamic coordination in concert with our Member States
and value-added partners to effectively manage the newly emerging Zika virus epidemic,
with all of its continuing challenges. This ordeal clearly demonstrated Pan American
solidarity at its best.
The period under review, from mid-2015 through mid-2016, was filled both with
numerous achievements of which the Bureau can be justifiably proud as well as with
numerous challenges, some of which have been successfully resolved while others are
works in progress. Under the rubric of achievements, we responded vigorously and
provided strong leadership and dynamic coordination in concert with our Member States
and value-added partners to effectively manage the newly emerging Zika virus epidemic,
with all of its continuing challenges. This ordeal clearly demonstrated Pan American
solidarity at its best.
One of the most important lessons learned from the Zika experience was the
imperative need for solid and unequivocal public health action and communication, even
in the face of limited scientific evidence. We learned that in this delicate balancing act,
extra weight must be given to protecting the most vulnerable in ways that are best
supported by the available evidence. The Zika epidemic has so far vindicated this general
approach.
We also responded to at least nine nonoutbreak emergencies, including a severe
earthquake, an uncontrolled migrant situation, and a number of adverse weather–related
events. Though these events were concurrent in time with the Zika epidemic, we were
able to lead, coordinate, respond, and manage with agility—underpinned by the
collaborative support of our Member States and numerous partners. Our capabilities in
this area are anchored in the tremendous experience and expertise that we have built over
the past 40 years, both at the country level and at Headquarters, from lessons learned in
disaster response and management.
Our technical cooperation in the area of the International Health Regulations has
enhanced the core capacities of Member States. However, we recognize that the IHR
must be anchored within strong and resilient national health systems in order to guarantee
an effective emergency response.
Achieving strong and resilient health systems obligates ministries of health to
discharge the essential public health functions, especially those related to governance and
stewardship, while guaranteeing sustainable funding and universal reach. Human
resources must be well trained and fit for the purpose; adequate in numbers, including
reserves; and equitably distributed across geographic areas. The Secretariat remains
committed to helping its Member States achieve these goals in the near term.
Our elimination efforts regarding measles and vertical transmission of HIV and
syphilis progressed noticeably during the year in review. We refocused the dialogue on
health systems financing so as to be able to better present evidence and options to
Member States that take greater cognizance of their economic realities within the global
and regional financial context. We enhanced our technical cooperation in the area of NCD prevention and control by more closely examining the impact of NCDs on the
national GDP.
 We continued laying the foundation pillars for our future work with Member
States towards the health-related Sustainable Development Goals (SDGs) while working
through a recently constituted commission to generate evidence to address the social
determinants of health and the cross-cutting themes of gender, ethnicity, and human
rights. Promising new initiatives are under way to ensure that the Bureau and the Region
as a whole will take the necessary steps to measure and achieve the SDGs related to
health.
We continued laying the foundation pillars for our future work with Member
States towards the health-related Sustainable Development Goals (SDGs) while working
through a recently constituted commission to generate evidence to address the social
determinants of health and the cross-cutting themes of gender, ethnicity, and human
rights. Promising new initiatives are under way to ensure that the Bureau and the Region
as a whole will take the necessary steps to measure and achieve the SDGs related to
health.
Looking forward, we recognize that our work related to Zika virus will remain a
substantial part of our unfinished agenda for many years to come, or at least until a safe,
affordable, and efficacious vaccine is produced. Ongoing research will be needed to
better elucidate a number of issues, such as what may be other competent nonvector
modes of Zika virus transmission and its pathogenesis. The Zika outbreak also brought
into sharp focus the need for us to redouble our technical cooperation efforts in order to
markedly increase access to comprehensive sexual and reproductive health services in
PAHO Member States.
Our efforts to advance universal health through the removal of client-patient
payments at the point of service must be strongly supported not only by ministries of
health but also by partners in the multilateral community, as the perpetuation of these
practices can have a heavy negative impact on access to health services, particularly for
those persons who live under conditions of vulnerability.
 Another major challenge for both the Bureau and Member States going forward is
how best to counter the influence of those industries (tobacco, alcohol, ultra-processed
foods, and sugary beverages) that can potentially be affected by national efforts to
strengthen regulatory capacity. These industries, which are usually global conglomerates
that are financially powerful and politically influential, have sought to influence the
development of laws, standards, and regulations that could reduce NCD risk factors.
Another major challenge for both the Bureau and Member States going forward is
how best to counter the influence of those industries (tobacco, alcohol, ultra-processed
foods, and sugary beverages) that can potentially be affected by national efforts to
strengthen regulatory capacity. These industries, which are usually global conglomerates
that are financially powerful and politically influential, have sought to influence the
development of laws, standards, and regulations that could reduce NCD risk factors.
In conclusion, the Bureau has unequivocally demonstrated its added value to
Member State efforts to improve the health and well-being of their populations. We have
exhibited leadership in our technical cooperation not only in relation to our agreed work
programs, but also in responding to the many unanticipated emergencies with which we
were confronted during the 2015-2016 period. We have provided expert technical
guidance based on the experiences and the lessons learned over many decades, and we
have successfully mobilized additional resources in support of the emergency responses.
As we look forward to this new era of the Sustainable Development Agenda, we
will continue providing visionary leadership in public health; expanding our partnerships
that add value and extend the reach of our technical cooperation; and increasing our
engagement and dialogue with Member States in order to ensure that our work remains
focused, relevant, and responsive to their needs.