PAHO Ethics Review Committee (PAHOERC)
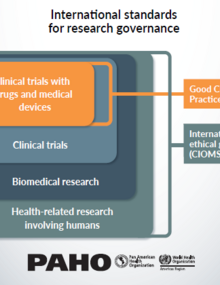
PAHOERC ensures that all research carried out with PAHO support, whether technical or financial, follows international standards for research with human participants. All research with human participants must also receive approval from an independent Ethics Review Committee (ERC) in the country where the study will take place. PAHOERC is the entity responsible for determining whether a proposal meets the definition of "research involving human participants." For more information, consult the PAHOERC Standard Operating Procedures (SOP).
PAHOERC is made up of a group of professionals from different disciplines, observers from key departments, and a Secretariat that is part of the Regional Program on Bioethics, within the Science and Knowledge for Impact Unit of the Department of Evidence and Intelligence for Action in Health (EIH/SK). Current PAHOERC members and observers are listed here.
PAHOERC 2026
:: Members
- Aleida Domingo Gisbert, Administrative Assistant, Strategic Fund for Public Health Supplies
- Bernardo Nuche Berenguer, International PAHO Consultant, HIV, Hepatitis, Tuberculosis, and Sexually Transmitted Infections
- Carla Saenz (Secretary), Regional Bioethics Advisor
- Carolina Chávez Cortes (External), Former International PAHO Consultant, Noncommunicable Diseases, Violence and Injury Prevention
- Lionel Gresh (Vice President), International PAHO Consultant, Viral Diseases
- María Paz Ade (President), Advisor, Malaria Diagnostics and Supply Management; Neglected, Tropical and Vector Borne Diseases
- Maristela Monteiro (External), former Senior Advisor, Alcohol and Substance Abuse
- Mauricio Beltrán Durán, Advisor, Blood Transfusion and Organ Transplants
- Omar Sued, Advisor, HIV/STI Care and Treatment; HIV, Hepatitis, Tuberculosis, and Sexually Transmitted Infections
- Pedro Ordúñez, Advisor, Chronic Diseases
- Regina Campa Sole, Advisor, Partnerships/External Relations, Partnerships and Resource Mobilization
- Sergio Surugi de Siqueira (External), Professor (Universidade Federal do Paraná); former member of Brazil’s National Research Ethics Committee (CONEP)
:: Observers
- Christina Marsigli, Department of External Relations, Partnerships and Resource Mobilization
- Gabriela Gergely, Ethics Office
- Nicole Wynter, Department of Planning, Budget and Evaluation
- Pamela Zúñiga, Office of the Legal Counsel
- Shirley Quesada, Department of Procurement and Supply Management
:: Secretariat
- Carla Saenz (Secretary), Regional Bioethics Advisor
- Magalys Quintana (Secretariat Support), Ethics Review Specialist
Submitting a proposal to PAHOERC
Proposals for review or consultation (see FAQs below) must be submitted through ProEthos, PAHOERC's portal for ethics review. After creating an account in ProEthos, follow the instructions in the platform to submit the proposal. Questions can be directed to PAHOERC@paho.org.
SEND PROPOSAL
NEW - See information about all the proposals received by PAHOERC from 2009 to date, including the title of the proposals, when they were submitted, the type of submission, the PAHO focal point and department responsible for the proposal, the country (or countries) where the proposal is intended to be carried out, and its current status.
PAHOERC INTRANET ACCESS
PAHOERC VIDEOS
Information Session
What needs ethics review during the pandemic?
Videos on Research Ethics
When does health research need ethics review?
What do Research Ethics Committees review?
External Resources
- Qualitative research and evaluation methods: integrating theory and practice, 4th Edition (Sage Publishing)
- Qualitative research methods for the social sciences 9th Edition (Amazon)
- Online courses on methods in social sciences (NEW)