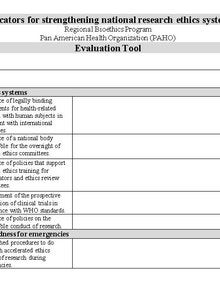
This document is an evaluation form developed by the Pan American Health Organization’s Regional Bioethics Program to assess and strengthen national research ethics systems. It outlines key indicators related to ethical standards for research involving human beings, including the existence of binding legal frameworks aligned with international guidelines, effective mechanisms for ethical oversight, national supervision bodies for ethics committees, and policies that promote ethical capacity-building for researchers and committees. The form also addresses transparency and research integrity through requirements such as prospective registration of clinical trials and policies for responsible conduct of research, as well as ethical preparedness for emergency situations, emphasizing procedures for accelerated ethical review during public health emergencies. |