Stakeholders from across the Caribbean countries participated in a two-day workshop, where the team shared presentations on the concept, MAP demonstration, and discussion among immunization stakeholders.
The MR-MAPs workshops aim to develop a partnership with national immunization stakeholders, to understand their perspective on the potential programmatic benefits of MR-MAPs' attributes in the context of the country's immunization barriers. In addition, it seeks to identify and examine critical criteria considered in the decision-making processes when comparing the current practice with the novel innovation, as well as the feasibility of the introduction and potential use cases of MR-MAPs.
The objectives are to determine country-specific immunization barriers relevant to MR delivery that MR-MAPs product attributes can address and to share the understanding and validating potential use cases for MR-MAPs: identifying critical product attributes for MR-MAPs and identifying data and evidence relevant to introduce MR-MAPs, but not deciding whether to introduce the product.
a
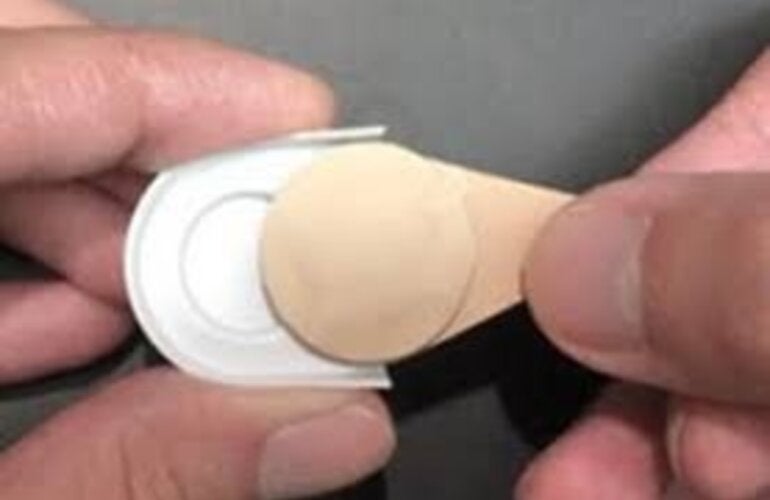
Microarray patches (MAPs) are a needle-free vaccine delivery technology consisting of hundreds or thousands of micro-projections on a 'patch coated with, or composed of, a dried vaccine. The vaccine is applied to the dermis, or epidermis, of the skin. Microarray Patches show measles and rubella vaccines (MR-MAPs) are easier to administer than needle-and-syringe (N/S) and be less burdensome on vaccinators and the immunization system given the avoidance of reconstitution, the reduced wastage due to their single-dose presentation, potential thermostability, reduced weight, and improved acceptability. As such, MR-MAPs can potentially overcome multiple programmatic barriers to immunization in low- and middle-income countries (LMICs) and transform how vaccines are delivered within immunization programs.
During the launch, Minister of Health, Honourable Dr. Frank Anthony, remarked that Guyana is ready to work with PAHO/WHO on the intervention of measles micro patches when it becomes available.
PWR, Dr. Codina, noted that when it becomes available, this new intervention will promote equity and access in some remote areas where logistics, including transportation and cold chain, are difficult.
Dr. Daniel Salas, Executive Chief of the Comprehensive Immunization Programme PAHO/WHO, noted that immunization coverage has decreased during the COVID-19 pandemic in countries in the Americas. Stakeholders must strengthen all efforts to increase the coverage to over 95%.
MR-MAPs were identified during the Vaccine Innovation Prioritization Strategy (VIPS) as a priority innovation for which the Alliance will engage to advance development, policy, and access. A series of consultations with immunization stakeholders in countries and regions were foundational to selecting MAPs as a priority innovation.
Before the workshop, the methodology used was a situational analysis conducted in Guyana with significant stakeholders to identify strengths, opportunities, and threats to the feasibility of introducing the vaccines.
PAHO will continue collaborating with stakeholders for new vaccine introductions to improve the strategies to increase coverage in all the regions.